Recently, Dr. Feng Lin from College of Engineering, Peking University, published a research paper entitled “Mechanical transmission enables EMT cancer cells to drive epithelioid cancer cell migration to guide tumor spheroid disaggregation” online in the journal Chinese Science: Life Science, revealing the regulating mechanism(s) of caner cells with mesenchymal-like phenotype within tumor tissue promoting epithelial cancer cell migration and invasion from the perspective of mechano-biology.
Credit: Science China Press
Recently, Dr. Feng Lin from College of Engineering, Peking University, published a research paper entitled “Mechanical transmission enables EMT cancer cells to drive epithelioid cancer cell migration to guide tumor spheroid disaggregation” online in the journal Chinese Science: Life Science, revealing the regulating mechanism(s) of caner cells with mesenchymal-like phenotype within tumor tissue promoting epithelial cancer cell migration and invasion from the perspective of mechano-biology.
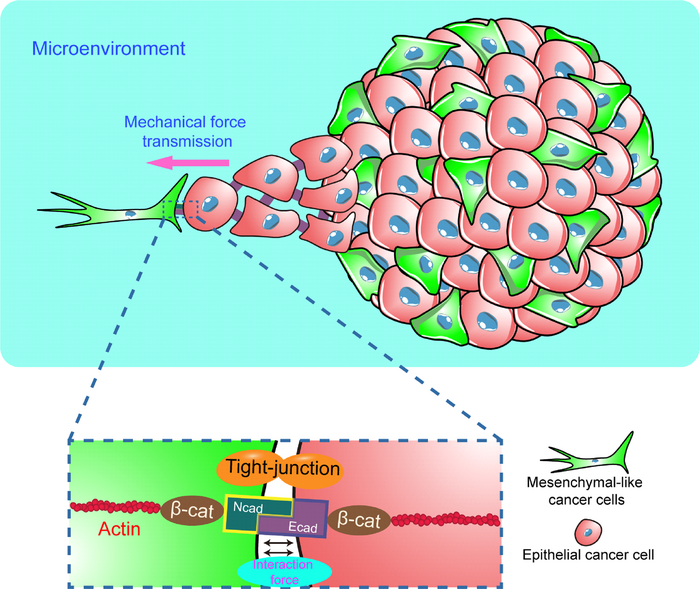
Firstly, the heterogeneous tumor spheroids mixed with epithelial and mesenchymal cancer cells were constructed using hanging drop method. It was found that the cells with mesenchymal-like phenotype preferred to dominate in the peripheral region during tumor spheroid self-organization. The authors found that mesenchymal-like cells migrated out of tumor spheroids faster and guided epithelial cancer cell migration and invasion through 2D dispersion and 3D invasion assays. This process is regulated by the enhanced contractility of tumor cells after EMT phenotypic switch, and is pertinent to the stiffness of microenvironment. It can effectively prevent the invasion of tumor cells by inhibiting cell contraction. The authors als found that N-cadherin on the surface of mesenchymal-like cells and E-cadherin on the surface of epithelial cells can form a heterophilic complex, which exercises the transmission function of mechanical force from mesenchymal-like cells to epithelial cells. Inhibiting the formation of intercellular N/E-cadherin heterophilic complex significantly reduce the interaction between mesenchymal-like cells and epithelial cells and the cancer invasion. In conclusion, this paper not only reports the key role of mechanical transmission between mesenchymal-like and epithelial cancer cells in mediating tumor cell migration and invasion, but also provides new clues and ideas for developing and tumor treatment strategy through regulating the mechanical interaction between heterogeneous cells within tumor tissue from the perspective of mechano-biology.
Zhang Qing, a Ph.D. student from Peking University, is the first author of this article. This research is supported by the National Natural Science Foundation of China and the China Postdoctoral Science Foundation.
See the article:
Mechanical transmission enables EMT cancer cells to drive epithelial cancer cell migration to guide tumor spheroid disaggregation
http://engine.scichina.com/doi/10.1007/s11427-021-2054-3
Journal
Science China Life Sciences
DOI
10.1007/s11427-021-2054-3




