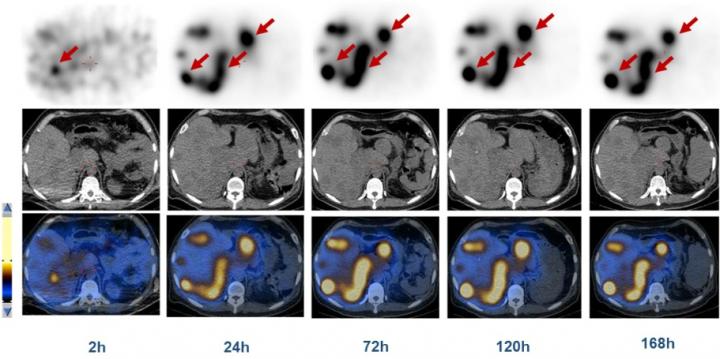
Credit: J Zhang et al., Peking Union Medical College Hospital, Beijing, China; X Chen et al., Laboratory of Molecular Imaging and Nanomedicine, NIBIB/NIH, Bethesda, MD
PHILADELPHIA – A first-in-human study presented at the 2018 Annual Meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI) demonstrates the benefits and safety of a new, long-lasting type of radionuclide therapy for patients with advanced, metastatic neuroendocrine tumors (NETs).
Lutathera-177 (177Lu)-DOTATATE (trade name Lutathera), a peptide receptor radionuclide tharapy (PRRT) with radiolabeled somatostatin analogues (peptides), was recently approved by the U.S. Food and Drug Administration for the treatment of NETs. It is the therapeutic part of a nuclear medicine theranostic pairing. Gallium-68 (68Ga)-DOTATATE is the diagnostic agent used in positron emission tomography/computed tomography (PET/CT) scans that first locates and marks the lesions for follow-up with targeted PRRT delivery directly to the tumor cells which express high levels of somatostatin receptors (SSTRs). Because the PRRT binds to receptors expressed by the tumor cells, healthy cells are unharmed.
However, the peptide quickly clears from the blood through the kidneys limiting the accumulation of radioactivity within tumors and making additional treatment cycles necessary to provide the therapeutic dose.
This first-in-human, first-in-class, Phase I trial (ID: NCT03308682) investigated the safety and dosimetry of a novel long-lasting radiolabeled somatostatin analogue that adds an albumin-binding Evans blue (EB, an azo dye) derivative to 177Lu-DOTATATE. Albumin, the most abundant plasma protein in human blood, is a natural transport protein and has a long circulatory half-life.
"177Lu-DOTA-EB-TATE is a "three-in-one" therapeutic compound, with an octreotate peptide to find the tumor, an Evans blue motif, which uses endogenous albumin as a reversible carrier to effectively extend the half-life in the blood and substantially increase targeted accumulation and retention within the tumor, and a therapeutic radionuclide to kill the tumor cells, to finally provide effective treatment of NETs," explains Shawn(Xiaoyuan) Chen, PhD, senior investigator, of National Institute of Biomedical Imaging and Bioengineering at the National Institutes of Health , Bethesda, Maryland.
For the study, conducted in collaboration with researchers at the U.S. National Institute of Biomedical Imaging and Bioengineering, 8 patients (6 men and 2 women ranging in age from 27 to 61 years old) with advanced metastatic neuroendocrine tumors were recruited from Peking Union Medical College Hospital and the Chinese Academy of Medical Sciences in Beijing, China.
Each patient underwent whole-body 68Ga-DOTATATE PET/CT. Five of the patients then accepted intravenous injection with a single dose of 0.35-0.70 GBq of 177Lu-DOTA-EB-TATE within one week, and were monitored at 2, 24, 72, 120 and 168 hours after 177Lu-DOTA-EB-TATE administration with serial whole-body planar and single photon emission computed tomography (SPECT)/CT images acquired. The other 3 patients accepted a dose of 0.28-0.41 GBq of 177Lu-DOTATATE and were monitored at 1, 3, 4, 24 and 72 hours with the same imaging procedures. Complete physical examinations, including vital signs, blood count, biochemistry, and immunology analyses were performed immediately before and 1, 3, and 7 days, as well as 3 months, after treatment.
Administration of 177Lu-DOTA-EB-TATE was well tolerated, with no adverse symptoms reported throughout the procedure and follow-up. The total effective dose equivalent and effective dose were 0.2048 ± 0.1605 and 0.0804 ± 0.0500 mSv/MBq for 177Lu-DOTA-EB-TATE and 0.1735 ± 0.0722 and 0.0693 ± 0.0317 mSv/MBq for 177Lu-DOTATATE. The liver, kidneys, bone marrow and total body received slightly higher doses (mGy/MBq) with 177Lu-DOTA-EB-TATE than with 177Lu-DOTATATE, while the spleen received lower doses with 177Lu-DOTA-EB-TATE. Blood clearance of 177Lu-DOTA-EB-TATE was also slower. Most importantly, 177Lu-DOTA-EB-TATE lasted in the tumors more than 4 times longer than 177Lu-DOTATATE.
Jingjing Zhang and Zhaohui Zhu of Peking Union Medical College Hospital point out, "By introducing an albumin binding moiety, this long-lasting radiolabeled somatostatin analogue has remarkably enhanced uptake and retention in SSTR-positive tumors, which is important to increase the therapeutic efficacy in patients. With proper selection of patients with advanced metastatic neuroendocrine tumors, 177Lu-DOTA-EB-TATE has great potential to be a highly effective treatment, while providing a safe dose with less frequency of administration than is possible with 177Lu-DOTATATE."
Abstract 118: "Safety, Pharmacokinetics and Dosimetry of a Long-lasting Radiolabeled Somatostatin Analogue 177Lu-DOTA-EB-TATE in Patients with Advanced Metastatic Neuroendocrine Tumors: A Phase 1 First-in-human Study," Jingjing Zhang, MD,PhD, Yuejuan Cheng, MD,Hao Wang, MD, Jie Zang, PhD, Fang Li, MD, Chunmei Bai, MD, and Zhaohui Zhu, MD, Peking Union Medical College Hospital; Gang Niu, MD, Orit Jacobson, PhD4, and Xiaoyuan Chen, PhD, U.S. National Institutes of Health, Bethesda, MD. SNMMI's 65th Annual Meeting, June 23-26, Philadelphia.
Link to Abstract
NOTE: See Abstract 2 of the SNMMI 2018 Annual Meeting on an improved PSMA-targeting agent for prostate cancer imaging and therapy, also an NIH study using Evans blue platform technology.
###
Please visit the SNMMI Media Center for more information about molecular imaging and personalized medicine. To schedule an interview with the researchers, please contact Laurie Callahan at 703-652-6773 or [email protected]. 2018 SNMMI Annual Meeting abstracts can be found online at http://jnm.snmjournals.org/content/59/supplement_1. Current and past issues of The Journal of Nuclear Medicine are online at http://jnm.snmjournals.org.
ABOUT THE SOCIETY OF NUCLEAR MEDICINE AND MOLECULAR IMAGING
The Society of Nuclear Medicine and Molecular Imaging (SNMMI) is an international scientific and medical organization dedicated to advancing nuclear medicine and molecular imaging, vital elements of precision medicine that allow diagnosis and treatment to be tailored to individual patients in order to achieve the best possible outcomes.
SNMMI's more than 16,000 members set the standard for molecular imaging and nuclear medicine practice by creating guidelines, sharing information through journals and meetings and leading advocacy on key issues that affect molecular imaging and therapy research and practice. For more information, visit http://www.snmmi.org.
Media Contact
Laurie F Callahan
[email protected]
@SNM_MI
http://www.snm.org
Original Source
http://www.snmmi.org/NewsPublications/NewsDetail.aspx?ItemNumber=29461