In a transformative leap for genome editing technologies, researchers have unveiled a breakthrough that surmounts a key obstacle in integrating large DNA sequences into mammalian genomes. Traditional methods relying on recombinases necessitate the use of large double-stranded DNA (dsDNA) donors, which unfortunately trigger severe innate immune responses. These immune reactions not only diminish integration efficiency but also severely restrict the practical use of these technologies, confining them mostly to ex vivo and immune-compromised environments. The new approach, termed INSTALL—short for integration through nucleus-synthesized template addition of large lengths—completely reimagines the nature of DNA donors used in recombinase-mediated genome editing.
INSTALL innovatively harnesses the mechanisms employed by integrative prokaryotic viruses and mobile genetic elements, demonstrating for the first time that recombinases can successfully work with circular single-stranded DNA (ssDNA) molecules designed to be ‘immune evasive.’ These circular ssDNA donors include a partially duplexed region that reconstructs the specific recombinase recognition site, thereby ensuring high-fidelity insertion. This molecular architecture not only optimizes recombinase compatibility but also dramatically reduces immune activation, a hurdle long standing in genome editing using dsDNA molecules.
One of the pivotal advantages of INSTALL lies in its ability to substantially diminish toxic innate immune responses in primary human cells—a feature that could redefine the boundaries of in vivo genome engineering. Unlike dsDNA donors, which can provoke immune pathways leading to inflammation and cell death, these circular ssDNA constructs slip under the immune radar. This immune stealth translates into markedly enhanced integration efficiencies and expands the clinical relevancy of recombinase-mediated DNA insertion, making systemic in vivo applications more feasible than ever before.
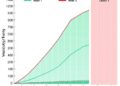
The researchers further demonstrated INSTALL’s versatility across a spectrum of system types, including both protein-guided and RNA-guided recombinases. This broad compatibility underscores INSTALL’s potential as a universal platform for kilobase-scale genome writing in human cells, a feat previously unattainable without viral vectors or extensive ex vivo manipulation. The ability to achieve precise, efficient, and large-scale DNA insertions opens up vast possibilities for therapeutic genome editing, synthetic biology, and functional genomics.
Perhaps one of the most eye-catching aspects of this study is the system’s performance when delivered in vivo via non-viral routes. Whereas lipid nanoparticle (LNP) delivery of dsDNA typically leads to dosage limitations due to immune toxicity, INSTALL-equipped ssDNA donors exhibit a significantly improved safety profile. This enhanced tolerability allows for broader dosing ranges, which is critical for effective systemic therapies targeting tissues beyond the liver and blood, including the central nervous system and solid organs.
The team’s experimental validation spans both primary human cell lines and mouse models, demonstrating that INSTALL can effectively evade immune detection while maintaining integration precision. These findings underscore the translational potential of the technique, suggesting that in the not-so-distant future, genome editing could be performed systemically in patients without triggering the deleterious immune sequelae associated with dsDNA use. Such advancements could drastically shorten timelines in gene therapy and significantly improve patient outcomes.
Beyond its immunological advantages, the molecular design of the circular ssDNA donors used by INSTALL intriguingly mimics naturally occurring viral integration processes. By reconstituting the recombinase recognition sequence within a partial duplex, the method cleverly exploits biology’s own toolkit for DNA integration. This synthetic biology-inspired strategy brings the benefits of viral mechanisms—potent integration and stable DNA insertion—without the inherent risks of viral vector use, such as insertional mutagenesis and immunogenicity.
Expanding on integration fidelity, INSTALL supports kilobase-scale DNA writing with high accuracy, a critical factor when designing therapeutic interventions or constructing complex genetic circuits. High-fidelity integration minimizes off-target effects, reduces the risk of unintended genomic instability, and ensures the precise engineering of desired phenotypes. This level of control is essential for clinical-grade genome editing where safety and efficacy are paramount concerns.
Moreover, the ability to introduce large DNA sequences efficiently enables the insertion of entire genes or regulatory elements, a capability that is transformative for diseases caused by large genetic defects or complex gene networks. INSTALL could feasibly facilitate the correction of hereditary disorders that require substantial genomic alterations, including Duchenne muscular dystrophy or cystic fibrosis. The method’s potential extends to sophisticated bioengineering tasks, such as installing synthetic metabolic pathways or enhanced immune cell receptors.
This technology also promises broad utility beyond therapeutic contexts. For instance, in fundamental research, INSTALL could empower scientists to more robustly manipulate mammalian genomes, paving the way for advanced functional screens and large-scale genetic studies. Its compatibility with multiple recombinase systems appears to make it adaptable to diverse research models, including those traditionally resistant to genetic modification due to immune sensitivity.
Crucially, INSTALL addresses an underappreciated but pervasive bottleneck in genome engineering: the balancing act between efficient DNA delivery and immune safety. By intersecting nucleic acid chemistry, virology, and protein engineering, this new methodology crafts an elegant solution that merges the stealth-like properties of circular ssDNA with the precise editing functions of recombinases. This synergy evidences a paradigm shift in how large-scale genome modifications can be safely and effectively executed in mammalian cells.
Looking ahead, the implications are vast. The technology’s ability to evade innate immunity and effect high-fidelity, large-scale DNA integration without viral vectors or toxic dsDNA molecules positions INSTALL as a foundation for next-generation gene therapies. As non-viral delivery techniques continue to evolve, combining them with immune-evasive templates could dramatically accelerate the clinical translation of complex genome editing platforms, minimizing adverse responses that have long hindered in vivo genetic interventions.
In conclusion, the introduction of INSTALL marks a milestone in genome engineering, unlocking the door to kilobase-scale genomic writing in living organisms without resorting to viral vectors or risking immune-mediated toxicity. By artfully blending the natural prowess of viral integration strategies with synthetic circular ssDNA constructs, researchers have devised a new path forward, promising safer, more efficient, and scalable genetic therapies and research tools. The scientific community eagerly awaits the broader applications and clinical translations that INSTALL will undoubtedly inspire.
Subject of Research: Genome editing technologies; recombinase-mediated integration; kilobase-scale DNA insertion; immune-evasive DNA donors; in vivo DNA delivery.
Article Title: Immune evasive DNA donors and recombinases license kilobase-scale writing.
Article References:
Tou, C.J., Xie, K., Ferreira da Silva, J. et al. Immune evasive DNA donors and recombinases license kilobase-scale writing. Nature (2026). https://doi.org/10.1038/s41586-026-10241-z
Image Credits: AI Generated
DOI: https://doi.org/10.1038/s41586-026-10241-z
Keywords: genome editing, recombinases, circular single-stranded DNA, immune evasion, kilobase-scale integration, non-viral delivery, innate immunity, synthetic biology, gene therapy
Tags: circular single-stranded DNA donorshigh-fidelity recombinase recognitionimmune activation avoidance in gene therapyimmune-evasive DNA editing toolsinnate immune response reduction in genome editingINSTALL genome editing methodkilobase genome editing technologylarge DNA sequence integrationprimary human cell genome editingprokaryotic virus-inspired DNA toolsrecombinase compatibility with ssDNArecombinase-mediated genome integration