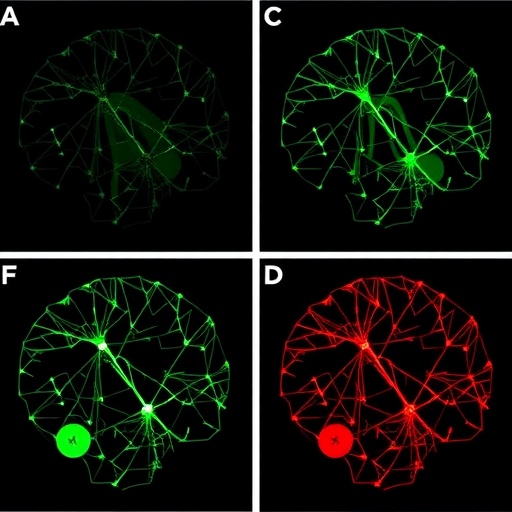
The human brain’s postnatal development is an intricate process involving a delicate orchestration of cellular growth, synaptic connectivity, and metabolic regulation. A groundbreaking study now shines a spotlight on a seemingly humble metabolic enzyme, glutamine synthetase (GS), positioning it as a pivotal regulator of cortical circuit maturation. This enzyme’s influence extends beyond simple metabolism, integrating with critical signaling pathways to shape astrocyte maturation and ultimately sculpt the neural networks essential for brain function.
Glutamine synthetase catalyzes the ATP-dependent amidation of glutamate to glutamine, a reaction fundamental to maintaining neurotransmitter cycling and amino acid homeostasis in the central nervous system. While GS’s neuroprotective roles in the adult brain are well-established, its precise function during early brain development has remained enigmatic. Leveraging innovative genetic techniques, researchers engineered mice with a selective ablation of GS in the cerebral cortex, illuminating the enzyme’s indispensable role during the postnatal window when brain circuits undergo rapid refinement.
Before birth, GS is robustly expressed in neural stem cells, priming the embryonic brain’s metabolic landscape. However, its expression shifts after birth, becoming predominantly enriched in astrocytes. These glial cells, which outnumber neurons, are essential for establishing and maintaining synaptic connections by modulating neurotransmitter availability and providing metabolic support. The selective deletion of GS in these cells did not perturb embryonic neurogenesis or the migration of neurons to their cortical positions, suggesting that GS’s developmental role is temporally and functionally distinct.
Postnatally, the absence of GS triggers profound astrocytic abnormalities. Astrocytes lacking GS exhibit stunted morphological development, characterized by reduced branching and diminished expression of developmental markers critical for their maturation. With time, these defective astrocytes adopt a reactive state, typically associated with neuroinflammation and a hallmark of various neuropathological conditions. This transition disrupts the delicate balance of neurotransmitter regulation and metabolic support required for optimal neuronal growth and synaptogenesis.
At the molecular level, GS deficiency impairs the homeostasis of amino acids, notably glutamine and glutamate, which profoundly affects cellular metabolism. A critical downstream consequence is the suppression of the mechanistic target of rapamycin (mTOR) signaling pathway within astrocytes. mTOR functions as a master regulator of cell growth, protein synthesis, and metabolic programming, and its activity is tightly linked to nutrient availability and cellular energy status. Without proper activation of mTOR, astrocytes fail to fulfill their metabolic roles, which cascades to impair neuronal dendritic arborization and synapse formation.
The disruption in astrocyte-to-neuron metabolic coupling manifests functionally as weakened neural activity within the cortex. This synaptic deficit correlates with behavioral phenotypes in the GS-deficient mice, including impaired motor coordination and altered social interactions. These features mirror symptomatology observed in numerous neurodevelopmental disorders, such as epilepsy and autism spectrum disorders, underscoring the clinical relevance of this metabolic pathway.
Remarkably, dietary supplementation of glutamine partially rescues both astrocytic maturation and synaptic deficits, highlighting the direct metabolic dependency of brain development on GS activity. This intervention not only restores metabolic equilibrium but also reactivates mTOR signaling, demonstrating a critical link between nutrient availability and cellular signaling mechanisms in guiding brain circuit formation.
The study’s revelations challenge the traditional neuron-centric view of brain development by positioning astrocytic metabolism and its regulation via GS as central to establishing functional neural networks. This metabolic perspective provides new conceptual frameworks for understanding neurodevelopmental disorders that may arise from subtle metabolic imbalances rather than purely genetic mutations.
Moreover, these findings open exciting therapeutic avenues. Modulating astrocytic metabolism through dietary or pharmacological means to enhance GS activity or downstream mTOR signaling could provide novel strategies for intervention in neurodevelopmental disorders characterized by synaptic and circuit dysfunction.
From a broader perspective, the research elegantly integrates molecular metabolism with cellular morphology and neural connectivity, illustrating the multifaceted roles of astrocytes beyond their classical supportive functions. The enzymatic activity of GS thus emerges as a linchpin connecting metabolic pathways to gene expression programs and structural maturation during a critical window of brain development.
Future investigations are poised to explore how GS-mediated metabolic control intersects with other pathways governing brain plasticity, and whether similar mechanisms operate in human neurodevelopment. Understanding these dynamics could deepen insights into the etiology of complex brain disorders and point toward new biomarkers and targets for early diagnosis and treatment.
In summary, glutamine synthetase sustains postnatal cortical circuit development by orchestrating mTOR-dependent astrocyte maturation and metabolic support for neurons. This discovery not only expands our molecular understanding of brain development but also highlights metabolism as a key driver of neural circuit assembly and function, with profound implications for neurodevelopmental disease research.
Subject of Research: Not applicable
Article Title: Glutamine synthetase sustains cortical circuit development via mTOR-mediated astrocyte maturation
News Publication Date: 2-Jan-2026
Web References: http://dx.doi.org/10.1093/procel/pwaf112
Image Credits: HIGHER EDUCATION PRESS
Keywords: Cell biology, glutamine synthetase, astrocyte maturation, mTOR signaling, brain development, neurodevelopmental disorders, metabolism, cortical circuits, synaptogenesis
Tags: astrocyte function in synaptic connectivityastrocyte-driven neural network formationastrocyte-neuron metabolic interactionscortical circuit formation mechanismsgenetic ablation of glutamine synthetaseglutamate-glutamine cycle in CNSglutamine synthetase role in brain developmentmetabolic regulation in neural stem cellsmTOR signaling in astrocyte maturationneuroprotective enzymes in early brainneurotransmitter homeostasis in cortical maturationpostnatal brain development enzymes