Inflammation plays a central role in the pathogenesis of numerous retinal diseases, notably dry age-related macular degeneration (AMD), one of the leading causes of vision loss worldwide. In these conditions, chronic inflammation triggers progressive retinal degeneration, leading to impaired visual function. Despite this well-recognized connection, clinical interventions that effectively quell inflammatory damage while simultaneously preserving the delicate neural architecture of the retina remain elusive. Conventional anti-inflammatory agents often fall short due to the complexity and chronicity of the inflammatory milieu, underscoring an urgent need for more nuanced therapeutic strategies that offer both immunomodulation and tissue neuroprotection.
In recent years, small extracellular vesicles (sEVs) derived from mesenchymal stem cells (MSCs) have emerged as an intriguing cell-free therapeutic platform. These vesicles, nanosized membrane-bound particles, carry bioactive molecules including proteins, lipids, and nucleic acids that can modulate recipient cell behavior. MSC-derived sEVs have demonstrated potential in various inflammatory conditions by exerting immunoregulatory functions and promoting tissue repair. However, the baseline therapeutic efficacy of unstimulated MSC-sEVs can be insufficient, particularly in environments characterized by high-grade inflammation such as the degenerating retina. Therefore, strategies to enhance the anti-inflammatory and neuroprotective cargo of sEVs have become a focal point of translational research.
In this context, recent investigations have focused on preconditioning MSCs with pro-inflammatory stimuli to potentiate the therapeutic payload of their secreted vesicles. Preconditioning with tumor necrosis factor-alpha (TNF-α), a pivotal cytokine in inflammation, has been shown to remodel the vesicle cargo, specifically enriching anti-inflammatory microRNAs (miRNAs) which are key post-transcriptional regulators of gene expression. A groundbreaking study has now elucidated the therapeutic dynamics of sEVs derived from TNF-α-preconditioned mesenchymal stem cells, hereafter termed T-sEVs, in the setting of inflammatory retinal injury.
Detailed molecular profiling revealed that TNF-α priming alters the miRNA content of sEVs in a way that skews the regulatory milieu toward anti-inflammatory pathways. Among the enriched miRNAs, miR-146a-5p stands out as a crucial modulator, renowned for its capacity to dampen inflammatory signaling cascades such as those mediated by toll-like receptors and nuclear factor-kappa B (NF-κB). The enriched presence of such miRNAs endows T-sEVs with a significantly enhanced ability to counteract macrophage-mediated inflammation, a key contributor to retinal degeneration.
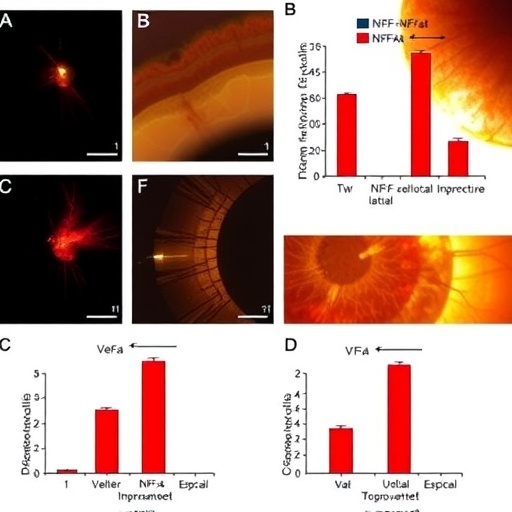
Functional assays demonstrated that T-sEVs are efficiently internalized by macrophages, the immune cells instrumental in orchestrating retinal inflammation. Once internalized, T-sEVs exert a pronounced inhibitory effect on the polarization of macrophages towards the pro-inflammatory M1 phenotype. This polarization suppression is accompanied by a reduction in the expression of hallmark pro-inflammatory genes such as Cd86, Il1r1, and Nos2, as well as a decrease in the secretion of inflammatory cytokines. This dual effect not only curtails the inflammatory cascade but may also preserve the retinal microenvironment conducive to neural survival.
The mechanistic underpinnings of these interactions were elucidated through integrated analyses combining vesicle miRNA profiling with recipient macrophage transcriptomics. These studies demonstrated direct targeting of inflammatory regulators, leading to modulation of critical signaling pathways including cytokine signaling and mitogen-activated protein kinase (MAPK) activity. This multilayered regulatory network highlights how T-sEVs reprogram immune cell behavior at the molecular level, offering a comprehensive attenuation of pathological inflammation.
To translate these findings into an in vivo context, the efficacy of T-sEVs was evaluated using a murine model of retinal degeneration induced by sodium iodate (NaIO3). Intravitreal administration of T-sEVs markedly preserved retinal function and anatomical integrity compared to controls. Notably, treated retinas exhibited reduced macrophage infiltration—a hallmark of diminished inflammation—and preservation of key retinal layers, as assessed by electrophysiological and histological analyses. These effects underscore the therapeutic potential of T-sEVs in mitigating inflammatory insult and neuronal loss in retinal degenerative conditions.
Equally important, thorough evaluations confirmed the safety profile of T-sEVs following local intravitreal administration. Both local ocular examinations and systemic toxicological assessments revealed no adverse effects, affirming that applying T-sEV-based therapy does not provoke harmful immune reactions or organ toxicity. This safety assurance is paramount for advancing such cell-free therapies toward clinical application.
Taken together, the evidence strongly supports the concept that inflammatory preconditioning of MSCs with TNF-α significantly enhances the therapeutic competence of their extracellular vesicles. T-sEVs emerge as potent immunomodulatory agents capable of rebalancing retinal immune homeostasis, suppressing detrimental macrophage activation, and protecting neural tissue integrity. This innovative approach offers a compelling alternative to cell-based therapies, circumventing challenges such as cell engraftment and potential tumorigenicity.
The implications of these findings extend beyond retinal disease, providing a paradigm for harnessing cytokine-primed extracellular vesicles for managing a spectrum of inflammatory disorders. By fine-tuning the bioactive cargo of sEVs through targeted preconditioning, it becomes possible to tailor their therapeutic properties to specific pathological contexts, maximizing efficacy while minimizing risks.
As this research enters the translational pipeline, several avenues warrant exploration, including optimization of dosing regimens, long-term efficacy studies, and evaluation in larger animal models. Furthermore, elucidating the interplay between sEVs and resident retinal cell types such as retinal pigment epithelial cells and Müller glia may unlock additional mechanisms of neuroprotection and regeneration.
In summary, T-sEVs derived from TNF-α-preconditioned mesenchymal stem cells present an innovative and promising cell-free therapeutic modality for inflammatory retinal degeneration. Their enriched anti-inflammatory miRNA cargo, effective modulation of macrophage polarization, and neuroprotective activity converge to address the complex pathology of diseases like dry AMD. Continued research and clinical development hold the promise of introducing a novel class of extracellular vesicle-based therapeutics that could revolutionize the management of debilitating retinal disorders that currently lack effective treatments.
Subject of Research: Not applicable
Article Title: Extracellular vesicles derived from TNF-α-preconditioned mesenchymal stem cells mitigate inflammatory retinal injury
News Publication Date: 17-Mar-2026
Web References: http://dx.doi.org/10.20517/evcna.2025.159
Image Credits: HIGHER EDUCATION PRESS
Keywords: Inflammation, Retinal degeneration, Mesenchymal stem cells, Small extracellular vesicles, TNF-α preconditioning, miR-146a-5p, Macrophage polarization, Cytokine signaling, MAPK pathway, Neuroprotection, Dry age-related macular degeneration, Cell-free therapy
Tags: anti-inflammatory therapy for retinal diseasescell-free therapies for retinal repairchronic inflammation in dry AMDextracellular vesicles from TNF-alpha primed MSCsimmunomodulation in retinal degenerationinflammation-induced retinal damage treatmentinnovative treatments for vision lossmesenchymal stem cell derived small extracellular vesiclesmolecular cargo of MSC-derived extracellular vesiclesMSC-sEVs enhanced by TNF-alpha primingneuroprotection in age-related macular degenerationtherapeutic strategies for retinal inflammation