When developing new drugs, understanding their effects on ion channels in the body, such as the human ether-a-go-go-related gene (hERG) ion channel found in neurons and heart muscle cells, is critical. Blocking hERG channels can disrupt normal heart rhythm, potentially leading to a fatal condition known as torsade de pointes. Current methods for assessing these effects typically involve invasive procedures like patch-clamp techniques or fluorescence microscopy. These methods alter cell properties and may affect measurement accuracy, requiring specialized equipment and expertise, which increases cost and complexity.
Credit: Daisuke Kawashima at Chiba University
https://pubs.rsc.org/en/content/articlelanding/2024/lc/d4lc00230j
When developing new drugs, understanding their effects on ion channels in the body, such as the human ether-a-go-go-related gene (hERG) ion channel found in neurons and heart muscle cells, is critical. Blocking hERG channels can disrupt normal heart rhythm, potentially leading to a fatal condition known as torsade de pointes. Current methods for assessing these effects typically involve invasive procedures like patch-clamp techniques or fluorescence microscopy. These methods alter cell properties and may affect measurement accuracy, requiring specialized equipment and expertise, which increases cost and complexity.
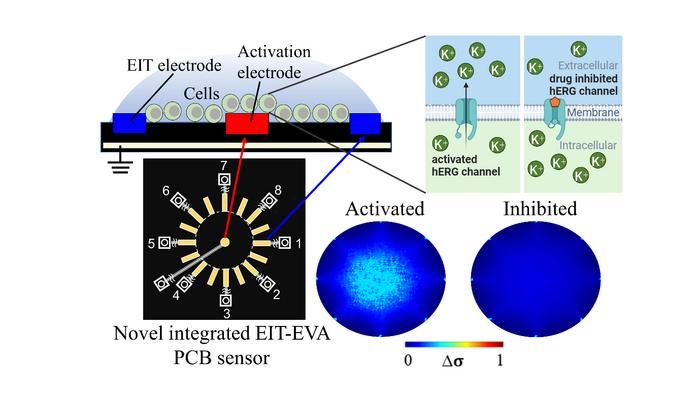
To address these challenges, researchers led by Daisuke Kawashima, an Assistant Professor at the Graduate School of Engineering at Chiba University, have proposed a novel, non-invasive method for real-time evaluation of drug effects on hERG channels. They developed a printed circuit board (PCB) sensor integrating electrical impedance tomography (EIT) with extracellular voltage activation (EVA). EIT measures impedance changes caused by ion movement, offering spatial information about extracellular ion distribution. EVA involves applying controlled extracellular voltages to induce changes in ion channel activity. This integrated approach allows researchers to non-invasively activate hERG channels and monitor real-time ion flow changes in response to drug exposure.
The study was published in the journal Lab on A Chip on May 23, 2024. It included contributions from Assistant Professor Songshi Li and Professor Masahiro Takei from the Graduate School of Engineering, Chiba University, along with Associate Professor Satoshi Ogasawara and Professor Takeshi Murata from the Graduate School of Science, Chiba University.
“This imaging technique is expected to serve as a new measurement and evaluation technology platform for medical and drug discovery,” says Dr. Kawashima.
The EIT–EVA PCB sensor is made from non-conductive epoxy glass fiber (FR-4 TG130) and measures 100 mm × 70 mm × 1.6 mm. It has 16 electrodes for EIT measurement arranged around a central activation electrode for EVA activation. Here’s how it works: The cells under investigation for drug effects on ion channels are placed on the sensor. A step voltage is applied to the activation electrode, which changes potential distribution in the extracellular medium surrounding the cells. This change affects the cell membrane potential, activating voltage-gated ion channels like the hERG channels. When these channels open, potassium ions move out of the cells, changing the extracellular resistance, which is measured by the EIT system.
The drug’s effect on the ion channel is then observed by monitoring changes in extracellular conductivity. If the hERG channels are not blocked by the drug, the concentration of potassium ions outside the cells increases quickly. However, if the drug blocks the channels, this increase slows down. The system calculates an inhibitory ratio index (IR) by measuring how fast the extracellular ion concentration changes over time, showing how much the drug inhibits the hERG channels.
To test their method, researchers exposed suspensions of genetically modified HEK 293 cells with hERG channels to the antiarrhythmic drug E-4031 at various concentrations (0 nM, 1 nM, 3 nM, 10 nM, 30 nM, and 100 nM). After mixing the drug and cell suspension into the sensor using a micropipette, they conducted baseline EIT measurements for 20 seconds to establish a baseline for ion movement. Subsequently, they alternated 20-second cycles of EVA activation and EIT measurements.
When the hERG channels were activated by EVA, the extracellular resistance decreased compared to the baseline (due to an increase in potassium ion concentration). However, as the concentration of E-4031 increased and blocked the hERG channels, fewer potassium ions were transported from the intracellular region to the extracellular region, resulting in a slower decrease in resistance.
From the IR response curve, the researchers found that the half-maximal inhibitory concentration or the concentration of drug required to inhibit hERG channel function by 50% was 2.7 nM. This value shows a strong correlation (R2 = 0.85) with results obtained from the established patch-clamp method, indicating close agreement between the two techniques. Compared to traditional methods in hERG channel screening, the proposed method is non-invasive, has a fast response time, and does not require special training. “This can lead to more efficient and shorter preclinical testing in the drug discovery process,” concludes Dr. Kawashima.
About Assistant Professor Daisuke Kawashima
Daisuke Kawashima received the B.S., M.S., and Ph.D. degrees in Mechanical Engineering from Tokyo Metropolitan University in Japan in 2012, 2014, and 2017, respectively. In 2017, he joined Chiba University in Japan as a Postdoctoral Fellow. He currently serves as an Assistant Professor at the Graduate School of Engineering. Prof. Kawashima has published over 40 research papers, which have been cited 250 times. His research interests include mass transfer in cells/tissues, electrical spectral analysis, and electrical tomographic imaging applied to biomedical and industrial fields.
Journal
Lab on a Chip
DOI
10.1039/d4lc00230j
Method of Research
Experimental study
Subject of Research
Cells
Article Title
Non-invasive hERG channel screening based on electrical impedance tomography and extracellular voltage activation (EIT–EVA)
Article Publication Date
23-May-2024
COI Statement
There are no conflicts to declare.