A groundbreaking study unveiled at the European Congress on Obesity 2026 in Istanbul, Turkey, has shed new light on the profound differences in how male and female bodies respond to obesity, potentially revolutionizing tailored medical interventions. Conducted by researchers at Dokuz Eylul University, this investigation rigorously analyzed the cardiometabolic and inflammatory profiles of nearly 1,134 adults living with obesity, definitively demonstrating that sex-specific physiological mechanisms critically shape disease trajectories associated with excess body weight.
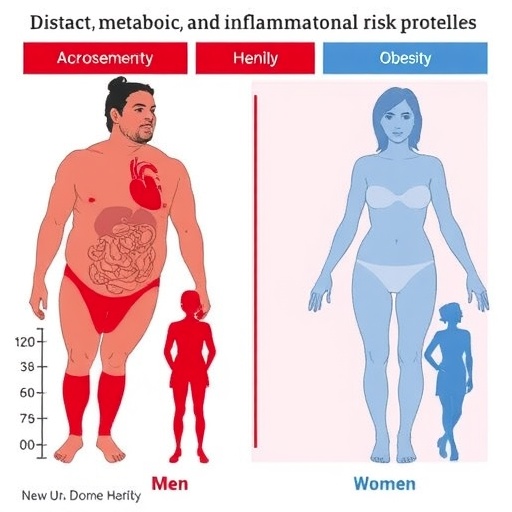
The research pinpointed a striking divergence in fat distribution and associated metabolic risks between men and women with obesity. Men exhibited a pronounced accumulation of visceral adipose tissue, the harmful fat surrounding internal organs, substantially increasing their vulnerability to metabolic syndromes, cardiovascular complications, and hepatic stress. This was corroborated by elevated liver enzyme levels—alanine aminotransferase (ALT) and gamma-glutamyl transferase (GGT)—which serve as sensitive biomarkers of hepatic dysfunction and chronic inflammation.
In stark contrast, women displayed a physiological overrepresentation of systemic inflammation and dyslipidemia. Their blood analyses revealed heightened levels of total cholesterol and low-density lipoprotein (LDL) cholesterol, both key contributors to the pathogenesis of atherosclerosis and type 2 diabetes. Accompanying these lipid anomalies were significantly increased markers of inflammation—erythrocyte sedimentation rate, C-reactive protein, and platelet counts—underscoring a distinctly inflammatory milieu potentially driven by immunological and hormonal factors unique to females.
These sex-specific variations are suggested to arise from complex interactions of hormonal regulation, immune system function, and adipose tissue biology. Estrogen, predominantly circulating in females, modulates subcutaneous fat deposition and immune response, promoting an inflammatory profile that may paradoxically increase cardiovascular risk despite a lower predilection for visceral fat accumulation. Conversely, testosterone and other androgens influence visceral fat expansion and metabolic enzyme activity, predisposing males to metabolic derangements and organ-specific damage.
Comprehensive anthropometric evaluations noted that while body mass index (BMI) was only marginally higher in men (37.5 kg/m² compared to 36 kg/m² in women), their waist circumferences—a proxy measure for visceral fat—were considerably larger, measuring 120 cm versus 108 cm in women. Elevated systolic blood pressure readings in men further corroborate their heightened risk for cardiovascular sequelae linked to central obesity.
From a biochemical perspective, men’s elevated liver enzymes and triglyceride levels signal a dysregulated hepatic metabolism which exacerbates systemic metabolic disturbances. Creatinine elevations, also noted, suggest potential renal strain likely secondary to these metabolic challenges. Women’s lipid profile abnormalities and inflammatory marker elevations denote a distinctly different pathophysiological pathway emphasizing systemic inflammation and altered lipid handling, potentially fueled by a stronger inherent immune response linked partly to gene dosage effects from the X chromosome.
This study’s strength lies in its robust dataset drawn from the Obesity Clinic at Dokuz Eylul University, incorporating a well-characterized cohort aged between 41 and 45 years on average, facilitating a granular assessment of metabolic and inflammatory parameters. However, the investigators prudently acknowledge limitations including the cross-sectional design precluding causal inferences, potential confounding variables, and the ethnic homogeneity of the Turkish population studied, which may limit extrapolation to diverse global populations.
The findings emphasize the necessity for clinicians to integrate sex-based biological distinctions into therapeutic paradigms to optimize obesity management, moving beyond one-size-fits-all approaches. Recognizing the differential risk profiles—men’s vulnerability to visceral fat-related metabolic and hepatic complications versus women’s propensity for cholesterol-driven inflammation—could catalyze the development of precision medical strategies that ameliorate disease burden more effectively.
Future research directions outlined by lead author Dr. Zeynep Pekel underscore the importance of longitudinal studies encompassing diverse ethnic groups to validate and extend these findings. In-depth molecular investigations into the hormonal regulation pathways and immune mechanisms underpinning these sex discrepancies will be pivotal. Moreover, correlating these biomarker profiles with longitudinal clinical outcomes can refine risk stratification and personalized treatment algorithms for people living with obesity.
The study dovetails with epidemiological data illustrating the burgeoning prevalence of metabolic syndrome globally, affecting over 1.5 billion adults by 2023. This syndrome embodies a cluster of cardinal risk factors including abdominal obesity, dyslipidemia, hypertension, and hyperglycemia, all conditionally modulated by sex-specific biology as now elucidated by this research. As metabolic syndrome underpins the majority of cardiovascular disease and type 2 diabetes cases worldwide, such sex-specific insights bear immense public health significance.
By illuminating the divergent paths through which male and female bodies interact with excess adiposity, this investigation opens new frontiers in understanding obesity’s heterogeneity. The precision medicine revolution in obesity care beckons, promising interventions tailored not only to an individual’s lifestyle and genetics but critically, to their biological sex—ushering in a new era of targeted, efficacious obesity management that mitigates cardiometabolic and inflammatory risks.
Subject of Research: Sex-specific cardiometabolic and inflammatory responses in adults living with obesity
Article Title: Distinct Sex Differences in Cardiometabolic and Inflammatory Health Risks Among Adults with Obesity—Insights from a Large Turkish Cohort
News Publication Date: 12-Apr-2026
Web References: https://drive.google.com/file/d/1ltkKYPFxwGsvEQSfr_S_OPr3I1rr7tsI/view?usp=sharing
References: [1] Worldwide trends in metabolic syndrome from 2000 to 2023: a systematic review and modelling analysis | Nature Communications https://www.nature.com/articles/s41467-025-67268-5
Keywords: Obesity, Sex differences, Cardiometabolic risk, Inflammation, Visceral fat, Cholesterol, Liver enzymes, Metabolic syndrome, Precision medicine, Hormonal regulation, Immune response
Tags: cardiometabolic risks and obesitycardiovascular complications of obesitydyslipidemia in female obesitygender differences in fat distributioninflammatory markers in obese womenmetabolic syndrome in obesityobesity and chronic inflammation biomarkersobesity-related liver enzyme elevationsex-based inflammatory responsessex-specific obesity risk profilestailored obesity medical interventionsvisceral adipose tissue in obese men