In a transformative leap for cancer diagnostics, a pioneering study by researchers at the University of California, Los Angeles (UCLA), in collaboration with global partners, has unveiled a cutting-edge deep learning-based method for virtual multiplexed immunostaining (mIHC). This innovative technology offers a rapid, accurate, and non-destructive alternative to conventional staining techniques, which are often labor-intensive and prone to inconsistencies. Published in the journal BME Frontiers, the breakthrough leverages the power of artificial intelligence to generate multiplexed immunostained images from label-free tissue sections, promising to redefine how pathologists assess vascular invasion in thyroid cancer.
Traditional immunohistochemistry (IHC) remains a cornerstone in oncology for identifying cellular markers critical to diagnosis and treatment planning. However, it requires physically staining separate tissue sections for each marker, such as ERG for endothelial cells or PanCK for epithelial cells, which can lead to increased sample consumption, elevated costs, and potential variability between sections. Even the advanced multiplexed IHC methods, though capable of simultaneous multi-marker staining, demand complex protocols and specialized instrumentation, limiting their accessibility within routine clinical pathology. These challenges have driven the search for more efficient, scalable solutions.
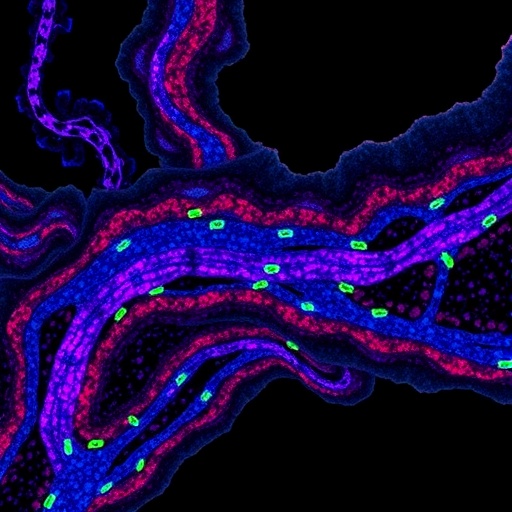
The UCLA-led team, spearheaded by professors Aydogan Ozcan and Nir Pillar, has developed a revolutionary approach that transcends traditional staining limitations by utilizing autofluorescence (AF) microscopy combined with advanced deep learning architectures. Their method captures unstained tissue images under multiple autofluorescence channels (DAPI, FITC, TxRed, and Cy5), providing rich intrinsic biochemical information without the need for exogenous dyes. This label-free imaging serves as the input for a conditional generative adversarial network (cGAN), which holistically synthesizes high-fidelity virtual IHC images encompassing ERG, PanCK, and classic hematoxylin and eosin (H&E) stains from the same tissue section.
At the heart of this system lies a cGAN framework composed of two neural networks working in tandem: a generator tasked with producing realistic virtual stains, and a discriminator that critically evaluates the authenticity of these images to refine the generator’s output iteratively. Enhancing the model’s multiplexing capability, the researchers incorporated a Digital Staining Matrix (DSM), a novel component concatenated with label-free inputs, enabling simultaneous generation of multiple marker images from a single input. This design eliminates the need for repeated physical staining procedures, preserving precious tissue material and expediting diagnostic workflows.
The team rigorously trained and validated their virtual mIHC model using a comprehensive paired dataset of autofluorescence and histochemically stained images collected from thyroid tissue microarrays. This extensive training allowed the cGAN to learn complex mappings between unstained autofluorescent signals and their stained counterparts, overcoming heterogeneity in tissue architecture and staining intensity. Quantitative assessments revealed that the synthetic images achieved remarkable concordance with traditional IHC slides in terms of cellular morphology, staining patterns, and marker localization.
To establish clinical relevance, blinded evaluations were conducted by board-certified pathologists who verified that the virtual stains faithfully replicated key diagnostic features of ERG and PanCK expression, as well as general tissue morphology via H&E staining. Importantly, the method demonstrated exceptional accuracy in identifying and localizing vascular invasion within thyroid tumor samples — a critical parameter linked to metastatic potential and patient prognosis. The pathologists noted the virtual stains preserved spatial context and cellular detail, essential for nuanced histopathological interpretation.
This virtual multiplexed immunostaining technology carries profound implications for both research and clinical practice. By obviating the need for multiple physical stainings, it mitigates the loss of tissue samples, reduces turnaround times, and lowers procedural costs. The AI-driven approach also circumvents variability inherent to manual staining protocols, enhancing reproducibility and diagnostic confidence. Moreover, its reliance on label-free autofluorescence images suggests easy integration with existing microscopy setups, facilitating deployment even in resource-limited settings.
Beyond thyroid cancer, the research team anticipates extending this framework to a wide array of tissue types and pathological conditions. Future studies are planned to validate performance across diverse multi-institutional cohorts, ensuring robustness and generalizability. The approach heralds a new paradigm for multiplexed histological analysis, where virtual staining powered by deep learning can augment or even replace conventional methods, leading to more personalized and timely patient care.
From a technical perspective, this work exemplifies the synergy between optical imaging and cutting-edge generative models. The cGAN’s capability to learn conditional mappings enables it to disentangle complex fluorescence signals and reconstruct multiple stains with high fidelity. The integration of the DSM further advances multiplexing by providing the network with explicit instructions about the desired stains, a strategy that can be adapted for other marker panels as the field evolves. This flexibility is pivotal for tailoring diagnostics to specific clinical questions.
In sum, the UCLA team’s deep learning-enabled virtual multiplexed immunostaining represents a watershed moment in digital pathology, combining precision, efficiency, and scalability. It opens a new vista for histopathology where AI augments human expertise, optimizes resource use, and sharpens diagnostic accuracy. As this technology matures, it promises to become a mainstay in pathology labs worldwide, catalyzing improved outcomes for patients confronting cancer and other diseases characterized by complex tissue microenvironments.
Subject of Research: Human tissue samples
Article Title: Deep Learning-Enabled Virtual Multiplexed Immunostaining of Label-Free Tissue for Vascular Invasion Assessment
News Publication Date: 10-Feb-2026
Web References: 10.34133/bmef.0226
Image Credits: Ozcan Lab@UCLA
Keywords
Deep learning, Immunohistochemistry, Artificial intelligence, Multiplexed immunostaining, Autofluorescence microscopy, Digital pathology, Generative adversarial networks, Thyroid cancer, Vascular invasion, Histopathology
Tags: AI-powered immunohistochemistrydeep learning for pathologydeep learning in cancer diagnosticslabel-free tissue imagingmultiplexed immunostaining without stainingnon-destructive tissue analysis methodsovercoming limitations of conventional IHCrapid multiplexed imaging techniquesscalable AI solutions in clinical pathologyUCLA cancer research innovationsvascular invasion assessment in thyroid cancervirtual multiplex immunostaining technology