In a groundbreaking study published recently in Nature Metabolism, researchers have unveiled a sophisticated molecular mechanism that sheds new light on hepatic insulin resistance (HIR) — a pivotal condition implicated in the pathogenesis of fatty liver disease, type 2 diabetes mellitus, and cardiovascular disorders. The study elucidates how oxidative modifications of a key enzyme, glycogen synthase kinase-3 beta (GSK-3β), radically transform its function in liver cells, disrupting the delicate balance between glycogen storage and glucose production. This novel insight not only deepens our understanding of HIR but also paves the way for the development of targeted therapeutic interventions designed to reverse or mitigate this metabolic dysfunction.
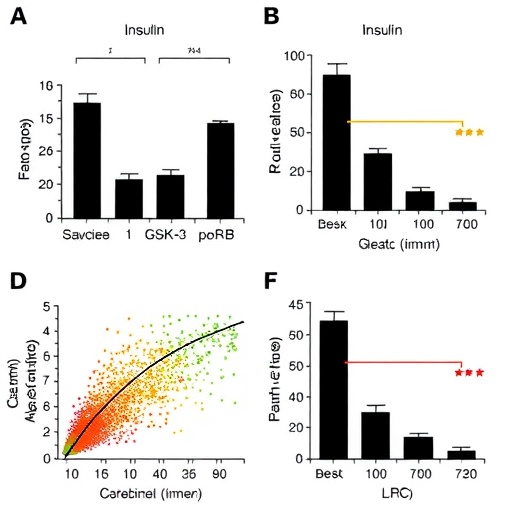
At the heart of this discovery lies the enzyme GSK-3β, historically recognized for its role in glycogen metabolism and insulin signaling. Under normal physiological conditions, insulin signaling inactivates GSK-3β through phosphorylation, specifically producing phosphorylated GSK-3β (p-GSK-3β), which ensures proper glycogen synthesis and controlled gluconeogenesis. However, the current research challenges this conventional paradigm by demonstrating that p-GSK-3β is not irreversibly inactivated but can be reactivated via a biochemical modification known as sulfenylation. This sulfenylation is driven by reactive oxygen species (ROS), molecules often elevated during metabolic stress.
The oxidative sulfenylation of p-GSK-3β represents a striking switch in its biological activity. Once modified, p-GSK-3β regains its kinase function and targets liver glycogen synthase, the enzyme responsible for converting glucose into stored glycogen. This phosphorylation event inhibits glycogen synthase, effectively terminating glycogenesis and halting glucose storage in hepatocytes. Consequently, there is a net effect of decreased glucose sequestration, a hallmark feature in the development of hepatic insulin resistance.
More remarkably, the sulfenylated p-GSK-3β extends its influence beyond glycogen synthesis. The study reveals that this reactivated kinase phosphorylates Forkhead box O1 (FoxO1), a transcription factor normally suppressed by insulin signaling. FoxO1, when released from insulin repression, translocates to the nucleus where it activates genes encoding gluconeogenic enzymes such as glucose-6-phosphatase and phosphoenolpyruvate carboxykinase. This dual action—terminating glycogenesis and promoting gluconeogenesis—creates a metabolic environment wherein hepatic glucose output remains inappropriately high despite insulin presence, embodying the essence of insulin resistance.
To substantiate these mechanistic insights, the researchers employed an array of cutting-edge techniques, including advanced mass spectrometry for the precise detection of sulfenic acid modifications, and genetically engineered liver organoids derived from human samples. These organoids faithfully recapitulated human liver physiology and pathophysiology, underscoring the clinical relevance of the findings. The conservation of this molecular mechanism in human liver tissue further validates its significance as a therapeutic target.
Importantly, this oxidative activation of p-GSK-3β signifies a previously unappreciated intersection between redox biology and insulin signaling. Reactive oxygen species have long been implicated in cellular damage and metabolic disease; however, this work demonstrates a nuanced regulatory role of ROS, whereby targeted sulfenylation fine-tunes enzyme activities with profound metabolic consequences. Such insights elevate the concept of oxidative post-translational modifications from a pathological byproduct to a dynamic signaling mediator.
Moving beyond fundamental science, this research opens exciting avenues for pharmacological intervention. Conventional therapies for hepatic insulin resistance and metabolic syndrome often yield limited success due to the complex pathway redundancies. By pinpointing the sulfenylated p-GSK-3β as a pivotal node controlling both glycogen synthesis and glucose production, novel drugs or molecular approaches designed to selectively inhibit this oxidized form of GSK-3β could restore insulin responsiveness and normalize hepatic glucose metabolism.
Furthermore, the study highlights the potential of antioxidants or redox-modulating compounds as adjunctive therapies. Targeting the ROS-sulfenylation axis could reduce the formation of enzymatically activated p-GSK-3β, thereby reasserting insulin’s capacity to suppress hepatic glucose output. This approach might complement existing treatments aimed at controlling hyperglycemia and enhancing insulin sensitivity.
Given the global burden of metabolic diseases, these findings are poised to have sweeping implications for public health. Fatty liver disease and type 2 diabetes affect hundreds of millions worldwide, driving morbidity and mortality. Understanding the molecular underpinnings of HIR at this unprecedented resolution allows clinicians and researchers to rethink strategies, moving toward more personalized and effective management.
The elegant dual-regulatory mechanism involving p-GSK-3β also calls for renewed exploration into how other oxidative modifications might control key metabolic enzymes under varying physiological and pathological states. This paradigm could extend to other organ systems and disease contexts, where oxidative stress and metabolic dysregulation intersect.
Moreover, the use of human-derived organoids as a platform for mechanistic studies exemplifies the potential of this technology to accelerate translational research. These microphysiological models provide a controllable, human-specific environment to dissect complex signaling networks and screen therapeutic agents, which can significantly reduce the reliance on animal models and streamline drug development pipelines.
Importantly, the researchers have provided a robust framework for future investigations to explore how sulfenylation dynamics are regulated in vivo and whether other cysteine residues within GSK-3β or related kinases may also be subject to oxidative control. This layer of redox regulation may operate as an essential sensor mechanism, linking metabolic stress with enzymatic output.
In considering the clinical landscape, these molecular insights offer hope for patients suffering from metabolic disorders unresponsive to current treatments. Therapeutically targeting the oxidized form of p-GSK-3β may circumvent the pitfalls of broader kinase inhibition, minimizing side effects while precisely addressing the root cause of hepatic insulin resistance.
Finally, this study exemplifies the power of integrating biochemistry, molecular biology, and clinical science to tackle one of the most pressing challenges in modern medicine. The revitalization of p-GSK-3β through cysteine sulfenylation has revolutionized our understanding of hepatic insulin resistance, transforming what was once considered an irreversible inactivated enzyme state into a dynamic modulator of liver metabolism.
As research advances, uncovering the full repertoire of redox-driven modifications and their impact on cellular signaling will be essential to design the next generation of metabolic therapies. This landmark study sets a new benchmark, offering a beacon of hope in the fight against diabetes, fatty liver, and cardiovascular diseases linked to insulin resistance.
Subject of Research: Molecular mechanisms underlying hepatic insulin resistance and the role of oxidative modifications of p-GSK-3β in regulating glycogenesis and gluconeogenesis.
Article Title: Revitalizing p-GSK-3β via cysteine sulfenylation promotes hepatic insulin resistance by differentially regulating glycogenesis and gluconeogenesis.
Article References:
Chen, J., Liu, Z., Gu, J. et al. Revitalizing p-GSK-3β via cysteine sulfenylation promotes hepatic insulin resistance by differentially regulating glycogenesis and gluconeogenesis. Nat Metab (2026). https://doi.org/10.1038/s42255-026-01507-x
Image Credits: AI Generated
DOI: https://doi.org/10.1038/s42255-026-01507-x
Tags: biochemical pathways in metabolic dysfunctioncardiovascular risk linked to hepaticcysteine sulfenylation in liver insulin resistanceinsulin signaling disruption in livermolecular mechanisms of hepatic insulin resistanceoxidative modification of p-GSK-3βoxidative stress in type 2 diabetesp-GSK-3β and glucose production imbalancereactive oxygen species and metabolic stressrole of GSK-3β in glycogen metabolismsulfenylation-driven enzyme reactivationtherapeutic targets for fatty liver disease