A groundbreaking new study has unveiled the devastating neurological consequences of a rare genetic mutation, shedding light on the intricate mechanisms that underlie microglial dysfunction and synaptic impairment. The mutation, known as CSF1R T567M, was investigated using cutting-edge patient-derived induced pluripotent stem cell (iPSC) cerebral organoids, offering an unprecedented window into the cellular pathology of CSF1R-related disorders. This research not only deepens our understanding of microglial biology but also paves the way for innovative therapeutic strategies targeting neurodegenerative diseases associated with microglial abnormalities.
Microglia, the brain’s resident immune cells, play a pivotal role in maintaining neuronal health, synaptic plasticity, and overall brain homeostasis. When microglia become dysfunctional, it can lead to a cascade of neuronal damage, ultimately compromising cognitive and motor functions. The T567M mutation in the colony stimulating factor 1 receptor (CSF1R) gene has been identified in patients exhibiting progressive neurological decline, yet the precise cellular and molecular consequences remained elusive until now.
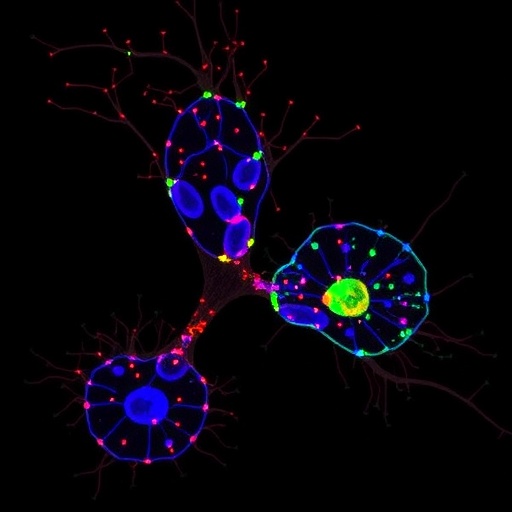
Employing iPSC technology, researchers generated cerebral organoids derived from patients bearing the CSF1R T567M mutation. This innovative approach allowed them to simulate human brain development in vitro, reproducing the complex cellular architecture and interactions observed in the living brain. Within these three-dimensional organoids, microglia were found to exhibit pronounced functional disturbances, characterized by impaired phagocytic activity and aberrant inflammatory signaling patterns.
One of the most striking findings was the disruption in synaptic organization and function within the patient-derived organoids. Synaptic integrity is crucial for effective neuronal communication, and the study revealed that CSF1R T567M microglia failed to support normal synaptic pruning and maintenance. This deficiency contributed to synaptic density abnormalities and deficits in neural network activity, mirroring the cognitive impairments experienced by patients afflicted with this mutation.
Intriguingly, the study delved into the signaling pathways downstream of the CSF1R receptor in microglia, revealing that the T567M mutation imposed conformational changes that hampered receptor phosphorylation and subsequent activation cascades. This molecular disruption led to altered expression of key genes involved in microglial survival, motility, and neuroprotective functions. These insights provide a mechanistic framework explaining how a single-point mutation in CSF1R can lead to widespread neuroinflammatory and neurodegenerative processes.
Moreover, the study highlighted the involvement of aberrant cytokine release profiles from mutated microglia, further aggravating neuronal stress and contributing to synaptic deterioration. Elevated pro-inflammatory mediators were observed alongside decreased anti-inflammatory signals, suggesting a tilt in microglial phenotypes toward a detrimental, disease-promoting state. These findings underscore the complex interplay between immune dysregulation and synaptic homeostasis in the context of neurodegeneration.
From a methodological standpoint, the use of patient iPSC-derived cerebral organoids represents a monumental leap forward in neurodegenerative disease modeling. Unlike traditional animal models, these organoids capture patient-specific genetic backgrounds, faithfully recapitulating human pathology at cellular and structural levels. This approach not only enhances the relevance of experimental outcomes but also offers a robust platform for personalized drug screening and precision medicine.
Further experimental investigations showed that correcting the T567M mutation via gene-editing technologies partially restored microglial functions and improved synaptic markers within the organoid model. This remarkable result hints at the potential for genetic therapies to counteract the deleterious effects of CSF1R mutations. However, substantial challenges remain in translating these laboratory successes into clinically viable treatments.
Importantly, this study accentuates the critical role microglial health plays in maintaining brain function, emphasizing that mutations affecting microglial receptors can have profound consequences beyond mere immune dysregulation. The neurodegenerative cascade initiated by CSF1R T567M mutation likely contributes not only to rare familial disorders but might also provide new perspectives on more common diseases such as Alzheimer’s and Parkinson’s, where microglial dysfunction is increasingly recognized as a major pathological factor.
The researchers call for intensified investigations into microglia-targeted therapies, advocating for the development of small molecules or biologics that can modulate CSF1R signaling or its downstream effectors. Such approaches might offer novel avenues to halt or reverse synaptic damage in affected individuals. The cerebral organoid platform described can serve as a critical testbed for these therapeutic innovations.
In summary, this landmark research elucidates how the CSF1R T567M mutation triggers microglial dysfunction and synaptic impairment, using a sophisticated patient-specific organoid model to reveal previously uncharted pathogenic mechanisms. This work represents a significant advance in neurobiology, offering hope that intervention strategies grounded in precise molecular understanding can eventually combat the debilitating consequences of CSF1R-related neurodegenerative diseases.
The implications of these findings extend well beyond the immediate disorder, opening new doors to comprehending microglia’s multifaceted roles in brain health and disease. By unraveling the molecular intricacies of how CSF1R mutations derail microglial regulatory functions and synaptic integrity, this study stands as a beacon guiding future research toward effective treatments aimed at preserving cognitive and neurological function in affected populations worldwide.
As the field rapidly advances, integration of patient-derived models combined with genome editing and high-throughput screening technologies promises a new era of personalized neuroscience. The comprehensive insights provided by this research underscore the excitement and potential surrounding microglial biology, particularly in the quest to unravel and treat complex brain disorders driven by immune-neuronal interactions.
With continued interdisciplinary efforts, the detailed mechanistic understanding gained from studies like this will almost certainly catalyze novel therapeutic paradigms, transforming how debilitating neurodegenerative diseases related to microglial dysfunction are diagnosed, managed, and ultimately cured in the near future.
Subject of Research: Microglial dysfunction and synaptic impairment caused by the CSF1R T567M mutation in patient-derived cerebral organoids modeling CSF1R-related neurodegenerative disorders.
Article Title: CSF1R T567M mutation induces microglial dysfunction and synaptic impairment in patient iPSC-derived cerebral organoids of CSF1R-related disorder.
Article References:
Chi, L., Tu, H., Li, Z. et al. CSF1R T567M mutation induces microglial dysfunction and synaptic impairment in patient iPSC-derived cerebral organoids of CSF1R-related disorder. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-02995-2
Image Credits: AI Generated
DOI: https://doi.org/10.1038/s41420-026-02995-2
Tags: 3Dcellular pathology of CSF1R mutationsCSF1R T567M mutation neurological impactgenetic mutations in microglial biologyinduced pluripotent stem cells for brain disease modelingmicroglia role in brain homeostasismicroglia-mediated synaptic plasticity impairmentmicroglial dysfunction in neurodegenerationneurodegenerative disease therapeutic strategiespatient-derived iPSC cerebral organoidsprogressive neurological decline mechanismssynaptic deficits in CSF1R disorders