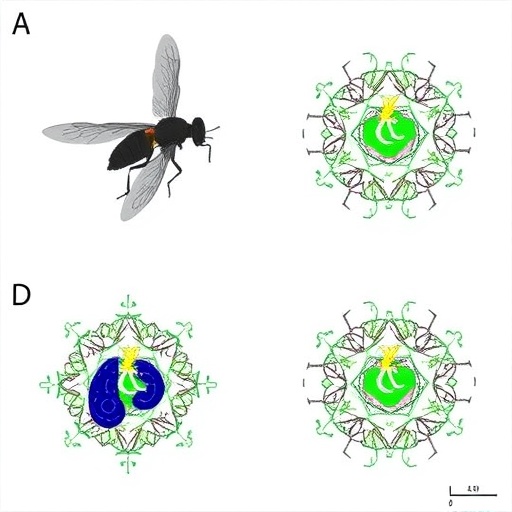
In an illuminating breakthrough that could reshape our understanding of aging and immune decline, researchers have uncovered a molecular mechanism by which immune function deteriorates with age, leading to gut dysbiosis and organismal vulnerability. The pivotal study, authored by Wang, Qi, Ma, and colleagues, delves into the intricacies of age-related immune senescence in Drosophila melanogaster, a powerhouse model organism for aging and immunity research. Their findings, published in Cell Death Discovery, expose a novel regulatory axis involving the cAMP response element-binding protein (CREB) and the peptidoglycan recognition protein SC2 (PGRP-SC2), revealing how CREB suppresses PGRP-SC2 expression to drive immune deterioration and gut microbiota imbalance during aging.
This work forms a cornerstone for deciphering the molecular crosstalk that governs immune competence in aging populations, with far-reaching implications for human health. The team’s elegant genetic and molecular dissection uncovers how CREB, a well-characterized transcription factor traditionally associated with neuronal plasticity and metabolism, exerts a repressive influence on PGRP-SC2 in the intestinal milieu. PGRP-SC2 is a crucial component of the innate immune system, responsible for recognizing microbial peptidoglycans and orchestrating appropriate immune responses to maintain gut homeostasis. The suppression of this immune sentinel by CREB creates a permissive environment for dysbiosis, a detrimental shift in the gut microbial community that exacerbates inflammatory stress and promotes physiological decline.
Aging is universally linked with immune senescence—a progressive weakening of immune defenses that undermines host resistance to infections and chronic inflammation. However, the molecular underpinnings that trigger and sustain this decline remained elusive until now. By leveraging the genetic toolbox offered by Drosophila, the study elucidates how age-dependent CREB activation functions as a master regulator of immune senescence. The researchers employed chromatin immunoprecipitation and reporter assays to demonstrate a direct binding of CREB to the promoter region of the PGRP-SC2 gene, leading to its transcriptional repression. This fine-tuning of gene expression underscores a tightly regulated checkpoint system that tilts immune homeostasis away from youthful resilience towards age-associated vulnerability.
The implications stretch further, as gut dysbiosis is increasingly recognized not merely as a bystander but as an active contributor to systemic aging and disease. The disruption of microbial equilibrium driven by CREB-mediated PGRP-SC2 suppression results in a hostile inflammatory environment within the gut. This chronic low-grade inflammation, often termed inflammaging, precipitates a cascade of physiological dysfunctions, including compromised barrier integrity and heightened susceptibility to pathogens. Notably, the study recapitulates aspects of human age-related gastrointestinal diseases, suggesting conserved pathways modulating immune-gut interactions across evolution.
Furthermore, the authors explored the temporal dynamics of CREB and PGRP-SC2 expression during the fly lifespan, uncovering a gradual increase in CREB activity concomitant with a decline in PGRP-SC2 levels. This inverse relationship mirrors the trajectory of immune decline and microbial instability, strengthening the causal link between these molecular events. The manipulation of CREB activity through genetic interventions yielded compelling results; flies with attenuated CREB signaling maintained robust PGRP-SC2 expression and exhibited prolonged gut health and immune competence, effectively delaying senescence phenotypes.
The discovery raises tantalizing prospects for therapeutic targeting. By intercepting the CREB-PGRP-SC2 regulatory axis, it may be possible to stave off or reverse premature immune aging and its sequelae. While translating findings from Drosophila to humans requires cautious optimism, the evolutionary conservation of CREB and PGRP functions bolsters the potential for analogous mechanisms operating in mammalian systems. The study paves the way for drug discovery efforts aiming to modulate CREB activity, potentially leveraging small molecules or gene therapy platforms to sustain gut immunity and microbiome integrity in elderly populations.
This research also broadens the conceptual framework linking transcriptional control with microbiota dynamics in the aging process. The integration of immunology, microbiology, and aging biology embodied in this work exemplifies the systems-level complexity inherent in senescence. By highlighting a transcription factor traditionally studied in neuronal contexts as a key driver of immune aging, the findings prompt a reevaluation of CREB’s functional repertoire and its systemic influence on organismal homeostasis.
Moreover, the study’s methodological rigor stands out, employing state-of-the-art genomic, microbiological, and physiological assays. The use of high-throughput sequencing and microbiome profiling techniques uncovered shifts in bacterial taxa correlated with CREB-PGRP-SC2 modulation, providing a detailed landscape of microbial alterations accompanying immune senescence. This integrative approach underscores the bidirectional relationship between host transcriptional networks and microbial ecosystems in shaping healthspan.
Importantly, the elucidation of CREB’s role in immune suppression challenges previous paradigms that predominantly considered immune decline as an irreversible consequence of aging. The reversibility demonstrated by genetic modulation introduces a revitalizing perspective that immune senescence may be a plastic and targetable process, opening avenues for interventions aimed at improving quality of life in aging individuals.
Beyond the scientific insights, this discovery holds profound implications for understanding age-associated pathologies such as inflammatory bowel disease, metabolic syndrome, and neurodegeneration, conditions often linked with immune dysfunction and microbiome imbalance. The interplay between CREB and PGRP-SC2 may represent a molecular nexus connecting these diverse disorders, offering a unified target for therapeutic exploration.
The research team’s findings also spark important questions regarding the upstream signals that trigger CREB activation during aging. Potential involvement of metabolic stress, oxidative damage, or hormonal changes in modulating CREB activity invites further investigation. Unraveling these initiating factors could deepen our grasp of the aging process and identify additional intervention points to preserve immune vigor.
In summary, the groundbreaking work by Wang and colleagues heralds a paradigm shift in our understanding of immune aging. The identification of CREB-mediated suppression of PGRP-SC2 as a driver of immune senescence and gut dysbiosis in Drosophila provides a mechanistic basis for age-related physiological decline and microbial imbalance. It offers a tantalizing glimpse into the molecular choreography orchestrating aging, immunity, and microbiota homeostasis. Future research inspired by these findings promises to unravel new therapeutic strategies aimed at enhancing healthspan by preserving immune function and microbiome stability across the lifespan.
Subject of Research: The molecular mechanism underlying age-related immune senescence and gut microbiota dysbiosis in Drosophila, focusing on the role of CREB and PGRP-SC2.
Article Title: CREB suppresses PGRP-SC2 to drive age-related immune senescence and gut dysbiosis in Drosophila.
Article References:
Wang, S., Qi, B., Ma, P. et al. CREB suppresses PGRP-SC2 to drive age-related immune senescence and gut dysbiosis in Drosophila. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-02955-w
Image Credits: AI Generated
DOI: https://doi.org/10.1038/s41420-026-02955-w
Tags: aging gut microbiota imbalancecAMP response element-binding protein functionCREB and age-related immune declineDrosophila as model for aging researchgenetic regulation of gut healthgut dysbiosis in Drosophilaimmune-microbiota interactions in aginginnate immune system in Drosophilaintestinal immune homeostasismolecular mechanisms of immune senescencePGRP-SC2 regulation in agingtranscriptional repression in aging immunity