A standard chemotherapy drug injures surrounding non-cancer cells, which can then awakens dormant cancer cells and promotes cancer growth, according to a new study publishing September 12th in the open access journal PLOS Biology by Ramya Ganesan of Emory University, US, and colleagues. The finding is important for understanding cancer recurrence and may point to important new targets to prevent it.
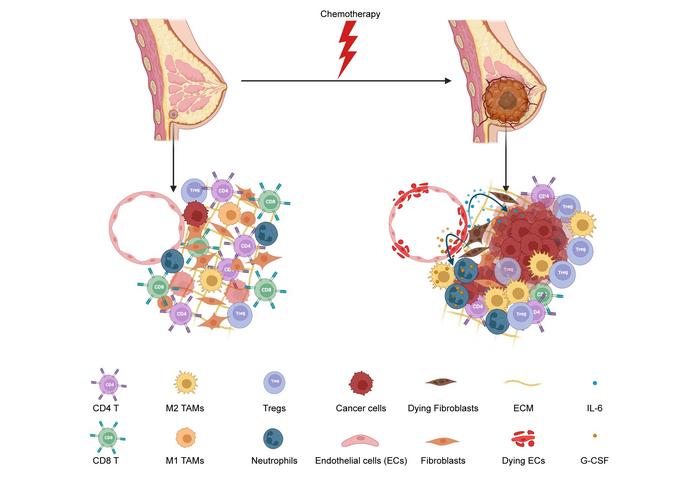
Credit: Ganesan R et al., 2023, PLOS Biology, CC-BY 4.0 (https://creativecommons.org/licenses/by/4.0/)
A standard chemotherapy drug injures surrounding non-cancer cells, which can then awakens dormant cancer cells and promotes cancer growth, according to a new study publishing September 12th in the open access journal PLOS Biology by Ramya Ganesan of Emory University, US, and colleagues. The finding is important for understanding cancer recurrence and may point to important new targets to prevent it.
Advances in cancer treatment, including chemotherapy, have dramatically reduced mortality for many types of cancer, including breast cancer. Nonetheless, up to 23% of breast cancer patients experience recurrence within the first five years. Treatment is meant to kill all cancer cells, but often, some cells enter a state of dormancy, in which they stop dividing and become unresponsive to chemotherapeutic agents. Recurrence occurs when dormant cells re-awaken and start dividing again.
Some studies have indicated that chemotherapy itself may promote escape from dormancy, but the mechanism of this effect has not been clear. To explore that question, the authors worked with both a cell model and a mouse model of breast cancer. Importantly, the cell model contained both cancer cells and non-cancer stromal cells, connective tissue cells that are found in breast and other tissue. They administered the chemotherapy drug docetaxel at physiologically relevant concentrations, and found that even at very low doses, stromal cells were injured, while cancer cells were not, and that treatment induced cell-cycle reentry in cancer cells.
The driver of this reawakening of dormant cells, the authors showed, was release of two key cell signaling molecules, granulocyte colony stimulating factor (G-CSF) and interleukin-6 (IL-6) by the injured stromal cells, which acted on the dormant cells to promote their growth, both in vitro and in vivo. That provided the team with potential anti-cancer targets, and they showed that antibodies that neutralized either G-CSF or IL-6, or a drug that blocked the mediator of those signals within cancer cells, inhibited awakening from dormancy due to docetaxel treatment.
These findings have several important implications. First, they highlight the importance of surrounding cells, not just the cancer cells themselves, in determining the response to chemotherapy. Second, they provide a possible mechanistic foundation for the observation that high serum levels of IL-6 are associated with early recurrence in breast cancer patients receiving chemotherapy, potentially strengthening the utility of that biomarker in planning treatment. Third, they provide new targets for preventing recurrence.
Dr. Ganesan and Dr.Sukhatme add, “Our paper highlights a deleterious effect of cancer chemotherapy: release of stromal IL-6 and G-CSF by taxane chemotherapy awakened dormant breast cancer cells, a postulated mechanism for tumor relapse. Transient blockade of cytokine signaling during chemotherapy administration may prevent tumor recurrence.”
#####
In your coverage, please use this URL to provide access to the freely available paper in PLOS Biology: http://journals.plos.org/plosbiology/article?id=10.1371/journal.pbio.3002275
Citation: Ganesan R, Bhasin SS, Bakhtiary M, Krishnan U, Cheemarla NR, Thomas BE, et al. (2023) Taxane chemotherapy induces stromal injury that leads to breast cancer dormancy escape. PLoS Biol 21(9): e3002275. https://doi.org/10.1371/journal.pbio.3002275
Author Countries: United States
Funding: This work was supported by startup funds provided to VPS, MKB and SSB by Emory University. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Journal
PLoS Biology
DOI
10.1371/journal.pbio.3002275
Method of Research
Experimental study
Subject of Research
Animals
COI Statement
Competing interests: I have read the journal’s policy and the authors of this manuscript have the following competing interests: VPS is on the SAB of BERG pharma and holds equity in Aggamin. MKB is in the SAB of Canomiks Inc and Anxomics LLC. SSB is a co-founder of Anxomics LLC. None of these companies are currently working in the cancer dormancy space.