In the rapidly evolving landscape of oncology, brain metastases represent a formidable challenge, particularly when they originate from clear cell renal cell carcinoma (ccRCC). A groundbreaking study recently published in Genes & Immunity sheds new light on the intricate immuno-molecular landscape of brain metastases in ccRCC patients, revealing novel insights that could redefine therapeutic strategies and improve patient outcomes. This comprehensive study, led by Roodhooft, Kinget, Mammone, et al., harnesses cutting-edge molecular techniques to dissect the complex interplay between tumor cells and the brain microenvironment, paving the way for innovative interventions in one of the most difficult-to-treat cancer contexts.
Brain metastases occur when cancer cells from a primary tumor in another organ migrate to the brain, establishing secondary tumors that disrupt neurological function and often contribute to a dismal prognosis. ccRCC, recognized as the most common subtype of renal cell carcinoma, is known for its aggressive nature and resistance to conventional therapies. The incidence of brain metastases in ccRCC has been rising, partly due to improved systemic therapies that prolong survival but fail to adequately penetrate the blood-brain barrier, thereby allowing microscopic disease to progress unchecked within the central nervous system.
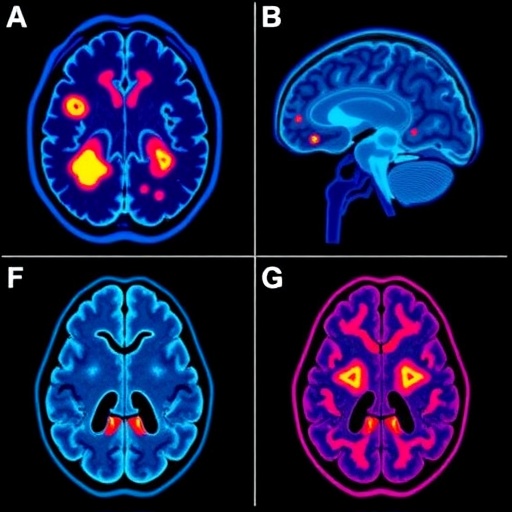
This investigation delves deeply into the immunological milieu surrounding brain metastases, a niche that has remained notoriously underexplored until now. Using state-of-the-art genomic and proteomic profiling, the researchers characterized the distinct immune cell populations and signaling pathways active within these metastatic brain lesions. Their findings uncovered an immunosuppressive environment dominated by regulatory T cells, myeloid-derived suppressor cells, and tumor-associated macrophages that collectively undermine anti-tumor immunity and facilitate immune evasion by ccRCC cells.
Significantly, the study identified differential expression patterns of immune checkpoint molecules such as PD-1, PD-L1, and CTLA-4 within the brain metastatic sites compared to primary kidney tumors. This revelation has immediate clinical relevance, suggesting that the efficacy of checkpoint inhibitors—a class of immunotherapy drugs—might differ substantially between primary and metastatic lesions. This insight could prompt oncologists to tailor immunotherapy regimens specifically for brain metastases, potentially incorporating combination approaches that counteract the unique immunosuppressive networks prevalent in the brain.
Moreover, the molecular characterization illuminated key signaling cascades dysregulated in brain metastases, including hypoxia-inducible factor (HIF) pathways, angiogenesis regulators, and metabolic adaptations favoring tumor cell survival in the hypoxic brain environment. These pathways represent promising targets for novel therapeutics, particularly small molecule inhibitors designed to disrupt the metabolic flexibility and vascular support systems that ccRCC brain metastases exploit. The integration of such agents with immunotherapies holds tremendous promise for synergistically enhancing treatment efficacy.
The blood-brain barrier (BBB), a formidable physiological barricade, presents a significant obstacle for drug delivery to brain metastases. Intriguingly, the analysis revealed alterations in BBB integrity and permeability associated with the metastatic microenvironment. Understanding these changes opens avenues for developing drug delivery systems capable of penetrating or transiently modulating the BBB, thereby increasing local drug concentrations in brain lesions without causing undue systemic toxicity. Nanoparticle-based carriers and focused ultrasound techniques are among emerging modalities that could capitalize on these insights.
Importantly, the study also tackled the evolutionary trajectory of ccRCC cells as they colonize the brain, uncovering genetic and epigenetic modifications that underscore phenotypic plasticity. Such plasticity endows the metastatic cells with the capacity to resist conventional treatments and adapt to the unique metabolic and immunological constraints of the central nervous system. This finding reinforces the necessity of dynamic treatment approaches that anticipate and overcome tumor adaptation, employing sequential or combination therapies based on real-time molecular monitoring.
Therapeutic resistance remains a cornerstone challenge in managing ccRCC brain metastases. By dissecting resistance mechanisms, including upregulation of drug efflux pumps, DNA repair enhancement, and immune evasion tactics, the research provides a framework for developing rational combinatorial regimens. These could include agents targeting DNA repair pathways, immune checkpoint inhibitors, and metabolic disruptors designed to sensitize tumors to therapy and prevent relapse.
Beyond molecular findings, the research underscores the heterogeneity of brain metastases both between patients and within individual lesions. This intratumoral heterogeneity complicates treatment but also offers an impetus for personalized medicine approaches. The deployment of single-cell sequencing technologies, as demonstrated in this study, enables the identification of subpopulations within tumors that may respond distinctly to therapies, guiding precision oncology strategies tailored to the biology of each patient’s disease.
Crucially, the clinical implications of these findings extend to predictive biomarkers capable of prognosticating response to emerging therapies. The identification of specific gene expression signatures and immune profiles predictive of treatment response will aid clinicians in selecting appropriate candidates for targeted therapies and immunotherapies, optimizing therapeutic success while minimizing unnecessary exposure to ineffective treatments.
This expanding knowledge base also has profound implications for clinical trial design. Future studies can now incorporate molecular stratification criteria for patient enrollment, ensuring that therapeutic efficacy is evaluated within biologically relevant subgroups. Additionally, the development of novel endpoints reflective of immunological and molecular changes rather than solely radiographic response may more accurately capture treatment benefits in brain metastases.
Another dimension of therapeutic innovation highlighted by this study relates to combinatorial strategies that integrate immunotherapies with radiation. Radiation has profound immunomodulatory effects that can potentially sensitize tumors to immune checkpoint blockade. Elucidating the precise molecular underpinnings of this synergy offers a roadmap for clinical protocols that maximize efficacy while managing toxicity.
The neuroimmune axis emerges as a critical frontier in the understanding of brain metastases biology. The intricate crosstalk between tumor cells and resident central nervous system immune cells, such as microglia and astrocytes, modulates disease progression and therapeutic response. The study’s findings that tumor-associated microglia adopt pro-tumoral phenotypes emphasize the need to explore this microenvironment as a therapeutic target, possibly reversing these immune cells’ role to support rather than hinder anti-tumor activity.
Moreover, the exploration of metabolic reprogramming in brain metastases reveals vulnerabilities that can be exploited pharmacologically. Brain metastatic cells exhibit altered glucose and lipid metabolism, shifting towards aerobic glycolysis and fatty acid oxidation pathways to meet the energetic demands of the metastatic niche. Targeting these metabolic pathways offers an innovative therapeutic angle, potentially disrupting the metabolic symbiosis between tumor cells and the brain microenvironment.
Given the poor prognosis traditionally associated with brain metastases, this study’s implications extend to improving quality of life and survival metrics for ccRCC patients. Enhanced understanding of molecular drivers and immune interactions sets the stage for developing therapies that not only extend survival but also mitigate neurological symptoms and preserve cognitive function, thereby significantly impacting patient care.
In conclusion, the research presented by Roodhooft et al. constitutes a landmark advancement in decoding the immuno-molecular complexity of brain metastases in clear cell renal cell carcinoma. By integrating multi-omic data, the study paves the way for next-generation therapeutic paradigms that merge immunotherapy, molecular targeting, and innovative drug delivery to surmount the unique challenges posed by brain metastases. As the oncology community continues to grapple with these clinical adversaries, such studies illuminate a promising path toward more effective, less toxic, and truly personalized cancer care for patients facing this daunting diagnosis.
Subject of Research: Immuno-molecular characterization and therapeutic strategies for brain metastases in clear cell renal cell carcinoma (ccRCC).
Article Title: Immuno-molecular features and therapeutic implications of brain metastases in clear cell renal cell carcinoma patients.
Article References:
Roodhooft, I., Kinget, L., Mammone, G. et al. Immuno-molecular features and therapeutic implications of brain metastases in clear cell renal cell carcinoma patients. Genes Immun (2026). https://doi.org/10.1038/s41435-026-00385-3
Image Credits: AI Generated
DOI: 07 March 2026
Tags: blood-brain barrier in cancer therapybrain metastases in renal cancerchallenges in treating brain metastasesclear cell renal cell carcinoma metastasisimmune insights in metastatic ccRCCimmune microenvironment of brain tumorsimmuno-molecular landscape of ccRCCinnovative interventions for ccRCC brain metastasesmolecular techniques in oncology researchnovel therapeutic strategies for brain metastasesprognosis of brain metastases in renal cancertumor-brain microenvironment interaction