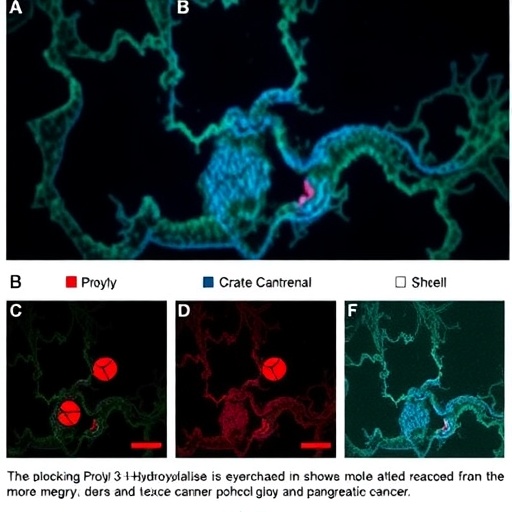
In the relentless battle against pancreatic cancer, a new beacon of hope has emerged from the laboratories of forefront cancer research. The enzyme prolyl 3-hydroxylase 1 (P3H1), an often overlooked participant in cellular biochemistry, has recently been spotlighted for its critical role in driving pancreatic tumor progression and modulating the immune landscape within the tumor microenvironment. The groundbreaking study authored by Bai, Liu, Fu, and colleagues, published in Nature Communications in 2026, unveils how targeting P3H1 can simultaneously thwart the aggressive advance of pancreatic cancer and reinvigorate macrophage-driven immunity, marking a significant breakthrough in cancer therapeutics.
Pancreatic ductal adenocarcinoma (PDAC), the most common form of pancreatic cancer, is notorious for its poor prognosis and resistance to conventional therapies. This malignancy’s lethality is compounded by a dense stromal matrix and an immunosuppressive microenvironment that inhibits the body’s natural defenses. Within this hostile milieu, P3H1 emerges as a pivotal enzyme implicated in post-translational modification of collagen and other matrix proteins, influencing extracellular matrix (ECM) stability and cellular communication in ways previously unappreciated.
At the molecular level, P3H1 catalyzes the hydroxylation of proline residues at the 3-position, a modification that distinctly alters collagen triple-helix stability. This biochemical action impacts not only the architectural integrity of the tumor stroma but also the dynamic crosstalk between cancer cells and infiltrating immune cells, particularly macrophages. Macrophages within the tumor microenvironment can adopt either tumor-promoting (M2-like) or tumor-suppressing (M1-like) phenotypes, meaning their functional state dramatically affects tumor growth and immune responsiveness.
The team’s meticulous investigations reveal that elevated expression of P3H1 in pancreatic tumors correlates with increased ECM rigidity and enhanced expansion of M2-like macrophages, creating conditions conducive to tumor progression and immune evasion. By employing genetic silencing techniques alongside small-molecule inhibitors specifically targeting P3H1, the researchers demonstrated a remarkable reversal of these malignant characteristics in preclinical models, underscoring the enzyme’s integral role in tumor biology.
Notably, the inhibition of P3H1 led to a marked decrease in collagen cross-linking and ECM stiffness, thereby mitigating the physical barriers that traditionally impede immune cell infiltration into the tumor core. This alteration in matrix composition facilitated a more permissive environment for M1-like macrophage activation, effectively reprogramming macrophages from a pro-tumorigenic to an anti-tumorigenic state. The shift was characterized by increased cytokine production linked to anti-tumor immunity and enhanced phagocytic capability against cancer cells.
These findings suggest that P3H1 is more than a structural enzyme; it is a master regulator of the tumor-immune microenvironment, orchestrating a symphony of biochemical and cellular events that determine tumor fate. The dual impact of P3H1 inhibition—targeting both matrix remodeling and macrophage polarization—affords a two-pronged therapeutic strategy, tackling tumor progression at its architectural and immunological cores.
Further exploration revealed that P3H1 inhibition did not compromise normal tissue homeostasis, highlighting its potential as a safe and selective target for drug development. The specificity of P3H1 inhibitors in disrupting tumor pathophysiology without eliciting deleterious systemic effects represents a monumental stride in precision oncology, especially for a cancer type that desperately needs innovative treatments.
Beyond the immediate therapeutic implications, this research provides profound insights into the intricate interplay between ECM remodeling enzymes and immune cell function in cancer. It challenges the dogma that structural enzymes are passive agents and promotes a reevaluation of the tumor microenvironment as an active participant in immune modulation and cancer progression.
The journey from basic enzymology to translational application exemplifies the progressive nature of biomedical science where understanding a single biochemical modification can unravel complex disease mechanisms. The authors’ work paves the way for integrating P3H1-targeted therapies with existing immunotherapies, such as immune checkpoint inhibitors, potentially overcoming the resistance that has plagued PDAC treatment.
This study also opens new avenues to investigate the role of P3H1 in other solid tumors given the ubiquitous nature of collagen and ECM remodeling in cancer biology. Could P3H1 modulation become a universal approach to enhance immune infiltration and disrupt tumor structure across malignancies? The tantalizing possibilities arising from this work underscore the need for expansive research into ECM enzymes as modulators of tumor immunity.
As the scientific community grapples with the complexities of cancer immunology, this study adds a crucial piece to the puzzle by illuminating how enzymatic activity shapes the tumor microenvironment at multiple levels. It emphasizes the delicate balance between tumor progression and the immune system, governed in part by biochemical modifications within the ECM, and highlights the potential to tip this balance therapeutically.
The implications of targeting P3H1 extend beyond therapeutic promise. They provoke deeper questions about how biochemical alterations in tumor matrix composition can either corrupt or support immune surveillance, and how the reconciliation of these processes could inspire next-generation approaches to cancer treatment.
The research by Bai and colleagues embodies the convergence of molecular biology, immunology, and biophysics, illustrating that subtle changes at the enzymatic level can have mosaic effects on tumor ecology. Targeting P3H1 hence reflects a sophisticated strategy that integrates multiple layers of tumor biology into a coherent, actionable framework for intervention.
Looking ahead, clinical translation of P3H1 inhibitors will require rigorous testing in human trials to validate efficacy and safety profiles. Equally important will be the development of biomarkers to stratify patients likely to benefit from such therapies and to monitor treatment response in real time.
In an era where immunotherapy is revolutionizing cancer care but often meets resistance in tumors like pancreatic cancer, the discovery of P3H1’s role offers a compelling avenue to overcome these hurdles. By dismantling the physical and immunological barricades erected by tumors, targeting P3H1 could refresh the armamentarium against one of the deadliest cancers known to medicine.
This transformative study not only enhances our molecular understanding of pancreatic cancer pathogenesis but also heralds a future where enzymatic targets within the tumor microenvironment redefine therapeutic landscapes. As research advances, P3H1 emerges as a potent symbol of hope—an enzyme whose inhibition might finally give pancreatic cancer patients a fighting chance for long-awaited remission.
Subject of Research: Pancreatic cancer progression and modulation of macrophage immunity via prolyl 3-hydroxylase 1.
Article Title: Targeting Prolyl 3-hydroxylase 1 inhibits pancreatic cancer progression and macrophage immunity.
Article References:
Bai, P., Liu, C., Fu, C. et al. Targeting Prolyl 3-hydroxylase 1 inhibits pancreatic cancer progression and macrophage immunity. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70452-w
Image Credits: AI Generated
Tags: cancer immunotherapy advancescollagen post-translational modificationsenzyme targeting in oncologyextracellular matrix remodeling in tumorsimmune evasion in pancreatic cancermacrophage activation in cancerpancreatic cancer treatment strategiespancreatic ductal adenocarcinoma researchpancreatic tumor progression mechanismsprolyl 3-hydroxylase 1 inhibitionstromal matrix in pancreatic cancertumor microenvironment modulation