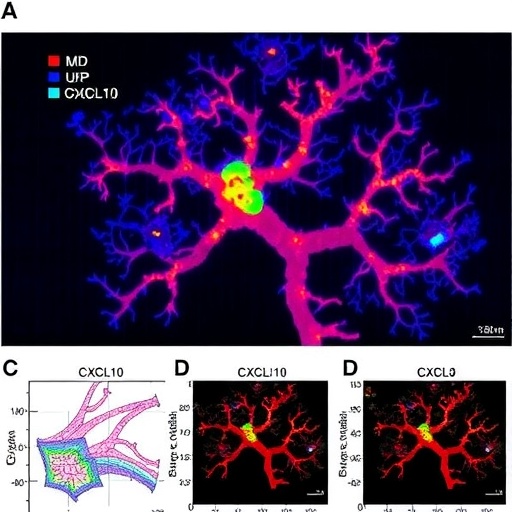
In a groundbreaking study published in Cell Death Discovery, researchers have illuminated a pivotal pathway by which astrocytes exacerbate brain injury following intracerebral hemorrhage (ICH). The work uncovers how astrocyte-derived CXCL10, a chemokine traditionally associated with immune responses, amplifies endothelial cell pyroptosis and disrupts the integrity of the blood–brain barrier (BBB) through a novel CXCR3/cGAS/AIM2 signaling cascade. This revelation sheds light on the molecular intricacies underlying secondary brain injury post-hemorrhage and opens new avenues for therapeutic intervention targeting neurovascular inflammation.
Intracerebral hemorrhage, a devastating subtype of stroke characterized by bleeding within brain tissue, often results in severe neurological deficits or death. One major contributor to the progression of injury is the breakdown of the BBB, a highly selective barrier that safeguards the central nervous system from harmful substances and peripheral immune cells. However, the mechanisms driving BBB disruption post-ICH have remained elusive until now. Sheng and colleagues identified astrocyte-secreted CXCL10 as a crucial mediator exacerbating endothelial dysfunction and cell death, thereby compromising BBB integrity.
The study pivots around pyroptosis, a regulated form of inflammatory cell death distinct from apoptosis, distinguished by the activation of inflammasomes and subsequent secretion of proinflammatory cytokines. Endothelial cells, which line cerebral microvessels and constitute a vital component of the BBB, were observed to undergo pyroptosis triggered by heightened CXCL10 signaling. This not only precipitates barrier leakage but also fuels a vicious cycle of neuroinflammation and neuronal damage, magnifying brain injury.
.adsslot_G2FsqZTA1Y{width:728px !important;height:90px !important;}
@media(max-width:1199px){ .adsslot_G2FsqZTA1Y{width:468px !important;height:60px !important;}
}
@media(max-width:767px){ .adsslot_G2FsqZTA1Y{width:320px !important;height:50px !important;}
}
ADVERTISEMENT
Astrocytes, star-shaped glial cells essential for maintaining neuronal health and vascular homeostasis, were found to overexpress CXCL10 in response to ICH-induced inflammatory cues. The chemokine binds to its receptor CXCR3 on endothelial cells, initiating a downstream cascade involving the cytosolic DNA sensor cGAS and the inflammasome component AIM2. Activation of this pathway culminates in the assembly of an AIM2 inflammasome complex, which drives pyroptotic cell death and the release of inflammatory mediators.
This intricate signaling axis provides a compelling mechanistic link between neuroimmune signaling and vascular integrity, highlighting how glial cells can remotely orchestrate endothelial demise. The cGAS-STING pathway, traditionally studied for its role in antiviral defense and autoimmunity, here emerges as a key player in sterile inflammation following brain hemorrhage. The coupling of CXCR3 receptor engagement with cGAS-AIM2 inflammasome activation underscores a sophisticated molecular crosstalk that translates glial signals into endothelial fate decisions.
Notably, the investigators employed a combination of in vitro and in vivo models, including primary cell cultures and rodent hemorrhagic stroke models, enabling a comprehensive assessment of molecular and functional outcomes. Intervention studies utilizing pharmacological inhibitors and genetic knockdown approaches effectively attenuated CXCL10-induced pyroptosis, restoring BBB permeability and improving neurological function. These data advocate for the therapeutic potential of targeting components of the CXCL10/CXCR3/cGAS/AIM2 pathway to mitigate secondary injury after ICH.
The relevance of these findings extends beyond hemorrhagic stroke, as BBB disruption and pyroptosis are implicated in a myriad of neurodegenerative and neuroinflammatory disorders such as multiple sclerosis and Alzheimer’s disease. Understanding how astrocyte-derived signals modulate endothelial cell death pathways offers fresh insight into the cellular interplay that governs brain homeostasis and pathology. Moreover, selective modulation of pyroptosis could represent a transformative strategy not only to preserve barrier integrity but also to temper the destructive inflammatory milieu within the brain.
Detailed mechanistic characterization revealed that CXCL10 binding to CXCR3 prompts accumulation of cytosolic double-stranded DNA fragments within endothelial cells, which in turn activate cGAS. This enzyme catalyzes the synthesis of the cyclic dinucleotide cGAMP that triggers downstream signaling, culminating in AIM2 inflammasome formation. The assembly of AIM2 inflammasomes then facilitates caspase-1 activation, gasdermin D cleavage, and the execution of pyroptosis. This cascade represents a convergence of chemokine signaling, nucleic acid sensing, and inflammasome biology within the cerebrovascular niche.
Importantly, the study delineates temporal dynamics of CXCL10 expression and inflammasome assembly during the acute and subacute phases following hemorrhage. Initial astrocytic CXCL10 release precedes endothelial activation, suggesting a causative role that sets the stage for progressive barrier breakdown. Interventions timed to disrupt this axis show promise in curbing inflammation and improving clinical outcomes, which is critical given the narrow therapeutic window in stroke management.
From a translational perspective, these findings invite exploration of CXCL10 or CXCR3 antagonists, as well as cGAS and AIM2 inhibitors, as adjunct therapies in hemorrhagic stroke. Given the multifunctional roles of these molecules, highly selective targeting—or temporally controlled modulation—will be necessary to minimize off-target immunosuppression. Still, this research provides a scientifically robust rationale for such endeavors, supported by thorough experimental validation.
Beyond therapeutics, the study prompts a reevaluation of the neurovascular unit as a dynamically interactive system where glial cells communicate death signals to endothelial cells under pathological conditions. The classical view of the BBB as a passive barrier is supplanted by a paradigm emphasizing its susceptibility to active inflammatory circuits mediated by non-neuronal cells. This paradigm shift enhances our fundamental understanding of CNS injury and may inspire biomarker discovery to monitor BBB status and inflammasome activation clinically.
The convergence of chemokine biology, inflammasome science, and vascular neuroscience embodied in this study exemplifies the power of interdisciplinary research to unravel complex neuropathologies. Future investigations may expand on how other astrocyte-derived factors interface with different components of the vascular and immune systems during injury and repair. Moreover, the potential crosstalk between pyroptosis and other forms of regulated cell death in endothelium opens fertile ground for exploration.
In summary, Sheng et al.’s research delineates a previously unrecognized astrocyte-to-endothelium communication pathway that potentiates BBB disruption through CXCL10-dependent pyroptosis. This mechanistic insight not only advances our understanding of intracerebral hemorrhage pathophysiology but also highlights promising targets for intervention aimed at preserving neurovascular integrity and improving patient outcomes. As stroke remains a leading cause of death and disability worldwide, such innovative molecular discoveries offer critical hope for the development of life-saving therapies.
The implications of targeting the CXCL10/CXCR3/cGAS/AIM2 axis extend well beyond acute brain injury, potentially impacting chronic neurodegenerative disease treatment by modulating neuroinflammation and vascular health. Efforts to translate these findings into clinical trials will require rigorous assessment of safety and efficacy but are firmly grounded in the compelling preclinical evidence now established. This work sets a new standard for mechanistic stroke research and exemplifies a transformative leap in decoding the molecular dialogues that underlie devastating cerebrovascular events.
Subject of Research: Mechanisms of blood–brain barrier disruption and endothelial pyroptosis mediated by astrocyte-derived CXCL10 via the CXCR3/cGAS/AIM2 pathway after intracerebral hemorrhage.
Article Title: Astrocyte-derived CXCL10 exacerbates endothelial cells pyroptosis and blood–brain barrier disruption via CXCR3/cGAS/AIM2 pathway after intracerebral hemorrhage.
Article References:
Sheng, W., Wu, Z., Wei, J. et al. Astrocyte-derived CXCL10 exacerbates endothelial cells pyroptosis and blood–brain barrier disruption via CXCR3/cGAS/AIM2 pathway after intracerebral hemorrhage. Cell Death Discov. 11, 373 (2025). https://doi.org/10.1038/s41420-025-02658-8
Image Credits: AI Generated
DOI: https://doi.org/10.1038/s41420-025-02658-8
Tags: astrocyte-derived CXCL10astrocytes and neuroinflammationblood-brain barrier disruptionchemokines in strokeendothelial cell pyroptosisendothelial dysfunction in hemorrhagic strokeinflammatory cell death in the brainintracerebral hemorrhage brain injuryneurovascular inflammation therapiessecondary brain injury mechanismssignaling pathways in brain injurytherapeutic interventions for ICH