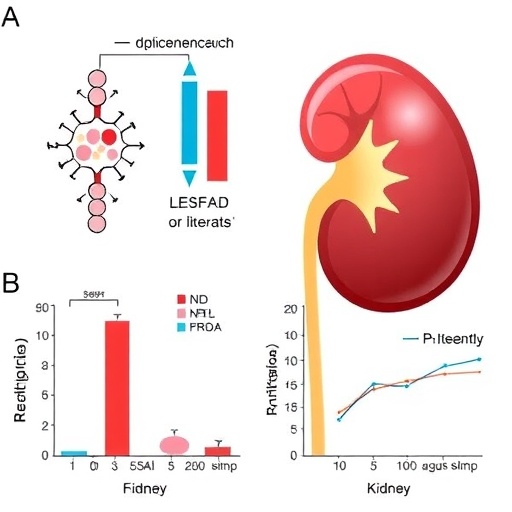
In a compelling advancement in the battle against sepsis-associated acute kidney injury (AKI), researchers have illuminated a novel therapeutic target capable of mitigating the devastating effects of this life-threatening condition. The study, conducted by Jia, Ji, Zhou, and colleagues, has revealed that inhibition of the protein BRD4, a pivotal player in gene regulation, significantly alleviates-sepsis-induced AKI by suppressing oxidative stress and inflammation driven by NOX4. This discovery propels forward our understanding of molecular mechanisms underlying AKI during sepsis and opens the door for potential pharmacological interventions.
Sepsis, a systemic inflammatory response to infection, frequently precipitates acute kidney injury, compounding mortality rates in critically ill patients. The pathophysiology of sepsis-associated AKI involves complex interplays between inflammatory cascades, oxidative stress, and cellular damage leading to renal dysfunction. Among the contributors to oxidative stress is NADPH oxidase 4 (NOX4), an enzyme isoform whose overactivation fosters excessive reactive oxygen species (ROS) generation, exacerbating injury and inflammation in renal tissues.
In their rigorous experimental framework, Jia et al. delved into the molecular crosstalk between BRD4 — a bromodomain-containing protein implicated in chromatin remodeling and transcriptional activation — and NOX4. They postulated that BRD4 acts upstream to regulate NOX4 expression, thereby modulating oxidative and inflammatory responses in sepsis-associated renal injury. Through pharmacological inhibition of BRD4, the researchers demonstrated a marked decrement in NOX4-mediated oxidative bursts within kidney cells under septic conditions.
The study employed a robust combination of in vivo and in vitro analyses to ascertain the therapeutic benefits of BRD4 suppression. In rodent models of sepsis-induced AKI, administration of BRD4 inhibitors conferred significant renoprotection, evidenced by improved biochemical markers of kidney function and histological preservation of renal architecture. These outcomes were tightly correlated with reduced ROS levels and dampened inflammatory cytokine profiles within renal tissues, underscoring the pivotal role of BRD4 in orchestrating deleterious oxidative-inflammatory signaling networks.
Mechanistically, the work revealed that BRD4 binds directly to promoter regions of the NOX4 gene, facilitating its transcriptional upregulation during septic insults. Blocking BRD4 disrupted this interaction, effectively downregulating NOX4 and attenuating downstream oxidative stress. This points to BRD4 not merely as a bystander but as a critical transcriptional regulator interlinking the epigenetic landscape with pro-oxidant pathways in kidney cells facing septic stress.
Moreover, the inflammatory milieu characteristic of sepsis involves cytokines such as TNF-α and IL-6, which exacerbate tissue damage and organ dysfunction. BRD4 inhibition was observed to significantly curb the release of these inflammatory mediators, suggesting its dual action in modulating both oxidative damage and inflammatory responses. This dual suppression signifies a multifaceted approach to combat the intertwined pathologies of sepsis-induced AKI.
Importantly, the therapeutic potential of targeting BRD4 extends beyond mere biochemical improvements. The suppression of oxidative and inflammatory pathways via BRD4 blockade translated into enhanced survival rates in septic animals, highlighting its promise for clinical translation. Such findings engender hope for the development of epigenetic modulators as adjunctive therapies in critical care settings where options for sepsis-associated AKI remain limited.
The implications of this study are profound, as they establish BRD4 as a master regulator in the injurious cascade of sepsis-driven kidney damage. Inhibition of BRD4 not only intercepts harmful genetic signaling but also orchestrates a reduction in pathological oxidative and inflammatory processes. This broad-spectrum mitigation could redefine therapeutic strategies, shifting focus toward epigenetic interventions that address root molecular dysfunctions.
Furthermore, the delineation of BRD4’s direct control over NOX4 expression underscores a novel axis in the pathogenesis of septic AKI. Previously, the therapeutic targeting of NOX4 itself has been contemplated; however, modulating its transcriptional regulation via upstream proteins like BRD4 offers a more refined, potentially safer means to attenuate oxidative stress without completely abolishing physiological ROS required for cellular signaling.
Translating these insights into clinical practice will necessitate comprehensive clinical trials to validate BRD4 inhibitors’ safety and efficacy in humans. Nonetheless, the existing data present a compelling case for the rapid advancement of these agents into translational pipelines. Given the urgent need for effective AKI therapies in sepsis, the unveiling of BRD4’s role is a beacon of scientific progress.
In essence, this groundbreaking research by Jia et al. accentuates the therapeutic value of epigenetic modulation in severe inflammatory and oxidative injury states. By targeting BRD4, clinicians may soon have a potent tool to counteract the multiple layers of molecular assault that characterize sepsis-associated AKI, ultimately saving lives and reducing the burden on intensive care resources worldwide.
This discovery also sparks broader inquiries into BRD4’s involvement in other organ injuries precipitated by systemic inflammation, paving avenues for future research beyond the kidneys. As the scientific community continues to decode the epigenetic language of disease, interventions like BRD4 inhibition emerge as promising candidates at the intersection of molecular biology and clinical medicine.
In conclusion, the work of Jia and colleagues marks a pivotal step in nephrology and critical care research. It challenges existing paradigms by demonstrating that targeted intervention at the level of transcriptional regulation can profoundly impact disease outcomes. BRD4’s inhibition represents not just a therapeutic mechanism but a conceptual leap toward epigenome-informed medicine for sepsis and beyond.
Subject of Research:
BRD4 inhibition as a therapeutic strategy for sepsis-associated acute kidney injury through suppression of NOX4-mediated oxidative stress and inflammation.
Article Title:
BRD4 Inhibition alleviates sepsis-associated acute kidney injury via suppression of NOX4-mediated oxidative stress and inflammation.
Article References:
Jia, J., Ji, K., Zhou, Y. et al. BRD4 Inhibition alleviates sepsis-associated acute kidney injury via suppression of NOX4-mediated oxidative stress and inflammation. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-03113-y
Image Credits: AI Generated
DOI: https://doi.org/10.1038/s41420-026-03113-y
Tags: acute kidney injury treatmentBRD4 and gene regulation in sepsisBRD4 inhibition in sepsischromatin remodeling in kidney diseaseinflammation in acute kidney injurymolecular targets for AKI therapyNADPH oxidase 4 and ROS productionNOX4 role in kidney injuryoxidative stress in sepsispharmacological interventions for sepsis-induced AKIsepsis inflammatory cascadessepsis-associated AKI mechanisms