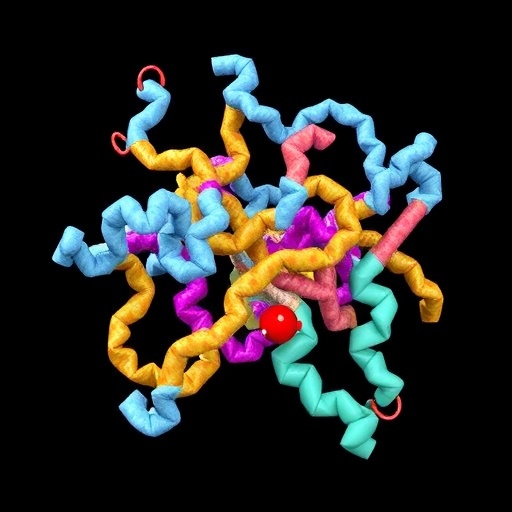
In a groundbreaking study that promises to revamp our understanding of cellular signaling, researchers have unveiled the intricate role of a tryptophan-rich allosteric network coupled with sodium dynamics in the activation of G protein-coupled receptors (GPCRs). These receptors, embedded in cell membranes, are pivotal in transducing extracellular signals into intracellular responses, governing physiological processes ranging from vision to neurotransmission and immune defense. The human adenosine A2A receptor (A2AR), a prototypical class A GPCR, served as the focal point for this investigation, revealing novel mechanistic insights into its functional modulation.
GPCRs represent the most extensive family of membrane receptors, underpinning myriad biological functions and comprising key targets of modern pharmacotherapy. Despite significant advances in drug development targeting GPCRs, the quest for selective, potent, and side-effect-minimized therapeutics remains elusive. This challenge stems predominantly from the dynamic allosteric nature of GPCR activation—a process characterized by conformational fluxes transmitted over long distances within the receptor, and between the receptor and the associated G proteins. Traditional structure-based approaches capture static snapshots but fall short in elucidating these subtle, yet pivotal, dynamic conformational transitions.
Addressing this critical gap, a multidisciplinary research team integrated cutting-edge rigidity theory, ^19F-NMR spectroscopy, and molecular simulations to decode the elusive allosteric communication pathways governing A2AR activation. Their investigative framework centered on Rigidity Transmission Allostery (RTA), an advanced mathematical model devised to trace mechanical signal propagation through protein structures. RTA analysis of the agonist-bound A2AR-G protein complex illuminated prominent allosteric conduits densely populated by tryptophan residues, accentuating their hitherto underappreciated role in receptor dynamics.
To substantiate these theoretical predictions, the team employed ^19F-NMR spectroscopy, harnessing the unique sensitivity of fluorine-labeled tryptophan probes to monitor conformational changes at both global and local scales. By targeting the TM6 transmembrane helix, a canonical reporter of receptor activation, alongside multiple fluorotryptophan sites dispersed throughout the receptor, they delineated the nuanced conformational ensemble shifts underpinning receptor activation and G protein engagement. This dual-level approach enhanced the granularity of the dynamic landscape, corroborating the integral participation of tryptophan residues in mediating allosteric signaling.
Further mechanistic insights emerged from comprehensive molecular dynamics simulations coupled with rigidity and geometry-based Monte Carlo methods. These simulations depicted how microswitch motifs and the conserved sodium-binding pocket undergo orchestrated conformational rearrangements during receptor activation. The Monte Carlo simulations, in particular, revealed transient intermediate states bridging inactive and activation-like conformations, offering a detailed roadmap of the receptor’s allosteric transitions hitherto unattainable by traditional experimental methods alone.
Central to the discovery is the characterization of a tryptophan-dense allosteric network that physically links the ligand-binding domain with intracellular signaling interfaces and the nucleotide-binding site of the coupled G protein. Contrary to the conventional view of tryptophans as mere structural scaffolds or NMR reporters, these aromatic residues emerge as critical transducers of allosteric information, orchestrating the relay of activation signals. The functional imperative of this network was underscored by mutagenesis experiments targeting the conserved toggle-switch residue W246 and the extracellular loop residue W143. These mutations precipitated widespread alterations in receptor conformational equilibria, reaffirming their central role in the allosteric circuitry.
In parallel, the research elucidated the mechanistic role of the conserved sodium pocket—a hallmark of class A GPCRs—in modulating receptor activation states. Experimental and computational analyses revealed that elevated sodium concentrations preferentially stabilize the inactive receptor conformations, whereas sodium egress facilitates the transition towards activation-prone ensembles. Critically, sodium depletion intensified the population of a precoupled receptor state, an intermediate configuration poised for G protein association, suggesting that sodium ion release operates as a molecular switch enabling signal propagation.
The implications of these findings extend well beyond the adenosine A2A receptor. Given the evolutionary conservation of the sodium pocket and key activation motifs across the class A GPCR superfamily, this study provides a unifying mechanistic framework to understand the dynamics of a vast array of medically pertinent receptors. This framework bridges ion-dependent modulation, microswitch dynamics, aromatic residue-mediated allostery, and receptor–G protein coupling, opening avenues for targeted pharmacological intervention tailored to receptor-specific allosteric landscapes.
A hallmark of this research is its integrative methodological architecture, combining experimental spectroscopy, rigid-body mathematical modeling, and molecular simulations. This synergy affords unprecedented resolution into GPCR conformational dynamics and signaling pathways, setting a new standard for structural and functional elucidation of membrane proteins. Furthermore, the generated experimental datasets and validated allosteric maps constitute invaluable training and benchmarking resources for emerging AI-driven protein dynamics prediction models, addressing a critical bottleneck in computational structural biology.
The potential applications of this approach are broad and transformative. Future extensions are envisaged to encompass other GPCR classes, diverse receptor–G protein pairings, pathophysiologically relevant receptor mutants, and ligand-specific biased signaling pathways. Such comprehensive datasets promise to refine drug discovery pipelines by facilitating the rational design of allosteric modulators that target specific conformational states, thereby enhancing therapeutic selectivity and reducing adverse effects.
Ultimately, this pioneering work exemplifies how the confluence of experimental biophysics, mathematical rigor, and computational power can demystify complex biomolecular processes. It heralds a future where the mechanistic underpinnings of receptor function are decoded with precision, enabling the design of next-generation therapeutics that manipulate receptor signaling with unprecedented fidelity and specificity. As the frontiers of molecular pharmacology advance, understanding the nuanced dance of tryptophan networks and sodium flux within GPCRs may transform how we conceive and engineer pharmacological interventions across a spectrum of diseases.
Subject of Research: Not applicable
Article Title: The Role of the Tryptophan-Rich Allosteric Network and Sodium Egress in GPCR Activation
News Publication Date: 2-Mar-2026
Web References:
DOI: 10.1073/pnas.2525502123
References:
Kitao et al., 2025, Proceedings of the National Academy of Sciences (PNAS)
Image Credits:
Kitao et al., 2025, Proceedings of the National Academy of Sciences (PNAS)
Keywords
GPCR activation, allosteric network, tryptophan residues, sodium pocket, rigidity transmission allostery, ^19F-NMR, molecular dynamics simulation, Monte Carlo simulation, receptor dynamics, G protein coupling, class A GPCR, allosteric modulation
Tags: ^19F-NMR spectroscopy in protein studyallosteric communication pathways in receptorscomputational modeling of membrane proteinsdrug target activation by allosteric networksG-protein coupled receptor signalingGPCR activation mechanismshuman adenosine A2A receptor modulationmultidisciplinary approaches in receptor biologyselective GPCR drug development challengessodium dynamics in GPCRsstructure-dynamics relationship in GPCRstryptophan-rich allosteric network