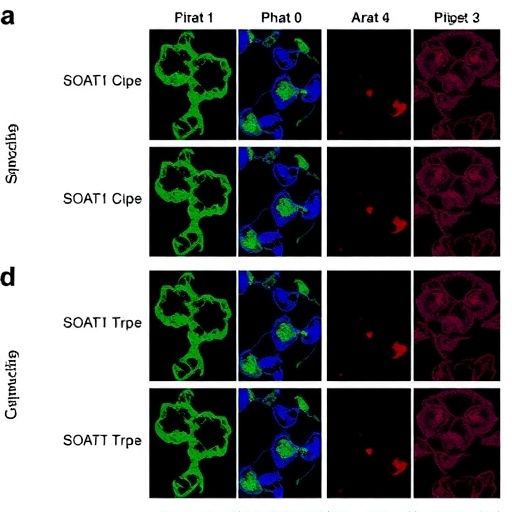
In a groundbreaking new study published in Nature Communications, researchers unveil a novel immune checkpoint pathway with profound implications for cancer therapy. The study zeroes in on SOAT1, known primarily as a sterol O-acyltransferase enzyme regulating cholesterol metabolism in cancer stem cells. This enzyme, now identified as an immune checkpoint, orchestrates regulatory T cell functions, revealing a previously uncharted mechanism by which cancer stem cells evade immune attack. Understanding and manipulating this pathway could revolutionize therapeutic strategies targeting the immunosuppressive environment surrounding tumors.
Cancer stem cells have long posed a significant challenge in oncology due to their notorious ability to resist conventional chemotherapies and evade immune surveillance. This investigation extends this knowledge by demonstrating how SOAT1 fosters immune privilege through an intricate trans-cellular communication axis involving 20(S)-Hydroxycholesterol and the G protein-coupled receptor GPR132. The meticulous work in murine models provides compelling evidence that inhibiting SOAT1 disrupts this axis, effectively impairing regulatory T cell functions and potentially restoring anti-tumor immunity.
Underpinning this discovery is the enzyme’s catalytic generation of esterified cholesterol derivatives, which act beyond mere metabolic byproducts. The researchers show that 20(S)-Hydroxycholesterol, a distinct oxysterol produced downstream of SOAT1 activity, serves as a signaling ligand for GPR132 on regulatory T cells. This cross-talk ensures the immunosuppressive microenvironment necessary for tumor persistence. By employing genetic knockouts and pharmacologic inhibitors specific to SOAT1, the study reveals a marked decrease in regulatory T cell-mediated immune suppression, leading to heightened immunogenicity of cancer stem cells.
The implications of this work reach far into the landscape of immuno-oncology. Regulatory T cells (Tregs) are essential modulators of immune homeostasis, often co-opted by tumors to blunt effective cytotoxic T cell responses. The demonstration of a metabolic-immune checkpoint mediated by SOAT1 integrates lipid metabolism with immune regulation, highlighting an axis that transcends traditional protein-protein immune checkpoint interactions such as PD-1/PD-L1 or CTLA-4. This novel pathway introduces an entirely new class of targets for immunomodulatory therapies.
Technically, the research team harnessed advanced murine tumor models that closely mirror human cancer stem cell biology to delineate the SOAT1-oxysterol-GPR132 pathway. Using flow cytometry, RNA sequencing, and lipidomics, they mapped how SOAT1 inhibition altered the molecular signature of regulatory T cells and their suppressive capacities. Importantly, the study did not limit observations to in vitro or ex vivo settings; in vivo analyses revealed that SOAT1 blockade reduced tumor growth and metastasis by reactivating adaptive immune responses, thus substantiating the therapeutic promise.
From a biochemical perspective, SOAT1 acts by esterifying cholesterol with fatty acids, effectively modifying the lipid composition within cancer stem cells. These lipid modifications were found to influence the secretion of bioactive lipid mediators, among them 20(S)-Hydroxycholesterol, which function as immune-modulating signals. The fine orchestration of lipid metabolism and immune function underscores a complex metabolic checkpoint that cancer cells exploit to shield themselves from immune clearance.
The G protein-coupled receptor GPR132, a less-explored player in immune regulation, emerges here as a critical receptor for the oxysterol signal. By binding 20(S)-Hydroxycholesterol, GPR132 triggers intracellular cascades that fortify regulatory T cell suppressive function. This newly characterized ligand-receptor interaction opens avenues for targeting GPR132 directly or its downstream signaling pathways, offering multiple layers for therapeutic intervention.
Beyond the immediate tumor microenvironment, this research suggests wider systemic implications for immune regulation via lipid metabolites. The interplay between cellular metabolism and immune checkpoints may represent a broader paradigm applicable to other diseases characterized by immune dysregulation. This could pave the way for metabolic reprogramming strategies in immunotherapy, exploiting the inherent plasticity of immune cells modulated by metabolic cues.
Consistent with the fantastical complexity of tumor immunobiology, the study also notes potential combinatorial strategies. SOAT1 inhibitors could synergize with existing immune checkpoint blockers or other metabolic modulators, amplifying anti-tumor immune responses while potentially mitigating adverse effects. Early preclinical data presented in the paper suggest that combining SOAT1 blockade with PD-1 inhibitors significantly improved tumor control, pointing toward a promising clinical translation.
The therapeutic promise is further underlined by the specificity of SOAT1 expression in cancer stem cells, contrasting with its limited expression in normal tissues. This selective expression profile minimizes the risk of global immune system disruption, enhancing the safety profile of SOAT1-targeted therapies. As a result, designing small-molecule inhibitors or RNA-based therapeutics against SOAT1 could offer a novel class of highly specific immunotherapies.
Moreover, the discovery spotlights the emerging importance of oxysterols in immune regulation, a field that has gained momentum in recent years. These cholesterol derivatives, long considered mere byproducts, are here elevated to pivotal signaling molecules that manipulate the balance between immune tolerance and activation. Such insights enrich our understanding of lipidomic regulation within the immune microenvironment, underscoring the intricate connections between metabolism and immunology.
The study also provides valuable tools and experimental frameworks for future research. The murine models developed for selective SOAT1 deletion and functional assays represent a significant technical advance, enabling the dissection of immune-metabolic pathways in vivo with unprecedented precision. This infrastructure will undoubtedly facilitate more detailed exploration of other enzymes and metabolites with immunomodulatory potential.
In summary, this seminal work expands the frontiers of cancer immunotherapy by revealing an unexpected metabolic checkpoint regulating regulatory T cells via cholesterol metabolism in cancer stem cells. The novel SOAT1-20(S)-Hydroxycholesterol-GPR132 signaling axis integrates metabolism and immunity, offering a compelling new target for enhancing anti-cancer immune responses. As the study transitions from bench to bedside, the prospect of metabolic checkpoint inhibitors adds an exciting dimension to the fight against cancer.
In essence, targeting SOAT1 and its downstream pathways could rewrite the narrative of immune evasion in cancer stem cells, potentially transforming the therapeutic landscape. The merging of metabolic regulation with immune checkpoint biology not only provides novel mechanisms but also opens a new frontier for precision immunotherapy. This innovation heralds a new era where small molecules once confined to metabolic functions become powerful modulators of immune landscapes.
The discovery of this trans-cellular signaling axis exemplifies the intricate communication between cancer stem cells and immune infiltrates. It underscores the necessity of viewing tumor biology through a multidimensional lens, where metabolism, immunity, and cellular signaling converge. This holistic perspective is critical for identifying vulnerabilities that can be exploited for durable and effective cancer treatments.
Looking ahead, further clinical evaluation and optimization of SOAT1 inhibitors will be required to harness this pathway fully. Researchers anticipate trials integrating metabolic checkpoint blockade with established immunotherapies. If successful, this approach could improve response rates, overcome resistance mechanisms, and ultimately lead to better clinical outcomes for patients with resilient cancers dominated by stem cell-like populations.
This research exemplifies the power of interdisciplinary collaboration, bridging immunology, cancer biology, and lipid metabolism. It highlights how sophisticated experimental designs and innovative thinking can uncover hidden layers of tumor-immune interactions, ultimately fostering the development of novel therapeutic paradigms with far-reaching impact.
Subject of Research: The molecular mechanisms by which inhibition of SOAT1 in cancer stem cells suppresses regulatory T cell function through a trans-cellular 20(S)-Hydroxycholesterol-GPR132 signaling pathway in mice.
Article Title: Inhibition of the cancer stem cell immune checkpoint SOAT1 suppresses regulatory T cell functions through a trans-cellular 20(S)-Hydroxycholesterol-GPR132 pathway in mice.
Article References:
Ding, Y., Fang, W., Xiang, R. et al. Inhibition of the cancer stem cell immune checkpoint SOAT1 suppresses regulatory T cell functions through a trans-cellular 20(S)-Hydroxycholesterol-GPR132 pathway in mice. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69305-3
Image Credits: AI Generated
Tags: 20(S)-Hydroxycholesterol signalingcancer immunotherapy targetscholesterol metabolism in cancer stem cellsdisrupting immunosuppressive tumor microenvironmentGPR132 receptor in immune regulationimmune evasion by cancer stem cellsoxysterol-mediated immune modulationregulatory T cell function inhibitionrestoring anti-tumor immunitySOAT1 immune checkpointsterol O-acyltransferase enzyme roletrans-cellular communication in tumors