The escalating prevalence of obesity, type 2 diabetes, and metabolic syndrome globally has precipitated a significant rise in fatty liver disease, positioning it among the most pervasive chronic liver conditions worldwide. The clinical spectrum of this disease has expanded to include metabolic dysfunction‐associated steatohepatitis (MASH), cirrhosis, and hepatocellular carcinoma, conditions that collectively pose daunting public health challenges. Central to effectively managing this broadening spectrum of metabolic dysfunction‐associated steatotic liver disease (MASLD) is the precise quantification of hepatic steatosis, a critical factor not only in diagnosis but also in monitoring therapeutic efficacy.
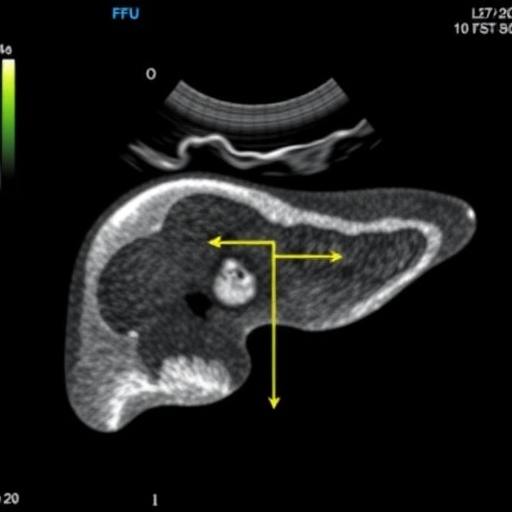
Historically, liver biopsy has been the benchmark for grading hepatic steatosis due to its direct histological evaluation. However, the invasive nature of biopsies, coupled with their inherent risks, high costs, and susceptibility to sampling errors, has underscored the urgent need for noninvasive diagnostic alternatives. Advanced imaging technologies such as magnetic resonance imaging-derived proton density fat fraction (MRI-PDFF) and proton magnetic resonance spectroscopy (^1H-MRS) have emerged as highly accurate, noninvasive modalities, yet their expense and limited accessibility restrict widespread clinical use. Recognizing these limitations, clinical researchers have turned to ultrasound-derived fat fraction (UDFF) as a promising, objective, and reproducible imaging technique that marries accessibility with precision in hepatic fat quantification.
A pioneering expert consensus led by Dr. Huixiong Xu and Dr. Hong Ding at Fudan University, China, responds to this clinical necessity by rigorously evaluating UDFF’s reliability, performance, and practical applicability. Published in the esteemed journal Portal Hypertension & Cirrhosis in March 2026, this comprehensive review synthesizes extensive multicenter trial data, cross-references with histopathological and MRI-based gold standards, and integrates expert clinical experience to provide a robust foundation for the systematic use of UDFF in routine practice.
The consensus study meticulously analyzed data from the largest multicenter cohorts to date, where UDFF measurements demonstrated strong validation against histopathology, MRI-PDFF, and ^1H-MRS. In parallel, the research benchmarked UDFF against established ultrasound grading systems, controlled attenuation parameter (CAP), and biochemical prediction models such as the fatty liver index and hepatic steatosis index. This multi-dimensional evaluation incorporated rigorous statistical assessments including correlation coefficients, intraclass correlation coefficients (ICCs), receiver operating characteristic (ROC) curve analysis, and Bland–Altman plots, all underscoring UDFF’s robust diagnostic capability.
Notably, UDFF exhibited exceptional reliability demonstrated by intra- and inter-operator ICCs of 0.94 or higher, indicative of high repeatability and reproducibility across operators and institutions. It showed strong correlations with histological steatosis grades, surpassing conventional imaging modalities in some instances, and even stronger concordance with MRI-PDFF data. Diagnostic accuracy was affirmed across multiple centers with area under the curve (AUC) statistics commonly above 0.90, reflecting outstanding sensitivity and specificity. A meta-analysis encompassing 1,150 patients further quantified these findings, revealing a pooled sensitivity of 90.4%, specificity of 83.8%, and a summary AUC of 0.93, with statistically low heterogeneity, consolidating UDFF’s clinical robustness.
To facilitate clinical decision-making, the consensus delineated provisional diagnostic thresholds derived from extensive datasets: 8% UDFF indicating significant steatosis (≥ S1), 14% reflecting moderate steatosis (≥ S2), and 20% identifying severe steatosis (S3). By adopting a dual-threshold strategy—defining rule-in and rule-out values—the approach refines diagnostic stratification and significantly reduces indeterminate cases, a key advancement especially pertinent in patients with higher body mass indices where imaging challenges prevail.
Further, evidence synthesized from pediatric studies and patients with concomitant conditions such as viral hepatitis, Wilson’s disease, and polycystic ovary syndrome highlighted UDFF’s versatility and reliable performance across diverse clinical contexts. This breadth of applicability suggests UDFF’s potential to become a standardized diagnostic tool within both adult and pediatric hepatology, pushing boundaries beyond traditional imaging techniques.
The implications extend well beyond diagnostic accuracy. UDFF’s noninvasive, quantitative approach offers operational simplicity, cost-effectiveness, and broad accessibility, positioning it as a feasible alternative to liver biopsy and expensive MRI modalities, particularly in low-resource healthcare settings. Its scalability could transform population-level screening, facilitate earlier detection of hepatic steatosis, and enable more effective monitoring of therapeutic responses in MASLD management.
While the consensus champions UDFF’s promise, the authors critically acknowledge existing limitations. Variability in reference standards and data paucity in certain subpopulations necessitate further large-scale, multicenter prospective studies employing unified diagnostic criteria to refine cutoff values and validate findings. Moreover, the clinical utility of UDFF in identifying high-risk MASH patients, especially those with advanced fibrosis, remains an area of active investigation to elucidate its full diagnostic and prognostic potential.
In summation, this expert consensus crystallizes UDFF as a reliable, accurate, and practical tool for the quantitative assessment of hepatic steatosis. By establishing standardized quality controls and proposing preliminary diagnostic thresholds, it lays the groundwork for broader clinical adoption, promising to enhance early MASLD detection, optimize therapeutic surveillance, and ultimately improve patient outcomes. As MASLD continues to burgeon as a leading global liver health challenge, UDFF offers a scalable, accessible solution aligning with contemporary clinical needs and technological advancements.
Subject of Research: Not applicable
Article Title: Chinese Expert Consensus on the Use of Ultrasound‐Derived Fat Fraction in the Assessment of Metabolic Dysfunction‐Associated Steatotic Liver Disease (2025 Edition)
News Publication Date: 6-Mar-2026
References: DOI: 10.1002/poh2.70042
Image Credits: Dr. Huixiong Xu and Dr. Hong Ding from Fudan University, China
Keywords: Metabolic Dysfunction‐Associated Steatotic Liver Disease, Ultrasound-Derived Fat Fraction, Hepatic Steatosis, Noninvasive Imaging, Diagnostic Accuracy, MRI-PDFF, Liver Biopsy Alternative
Tags: clinical guidelines for liver steatosis evaluationcomparison of liver biopsy and imaging modalitiesearly detection of metabolic dysfunction-associated steatohepatitisimaging techniques for fatty liver diagnosismetabolic dysfunction-associated steatotic liver disease assessmentMRI-PDFF limitations in fatty liver diagnosisnoninvasive hepatic fat quantification methodsrole of ultrasound in metabolic syndrome liver complicationstherapeutic monitoring of hepatic steatosisultrasound in metabolic liver disease monitoringultrasound-derived fat fraction for liver steatosis