In a groundbreaking new study set to reshape our understanding of amyotrophic lateral sclerosis (ALS), researchers have uncovered a crucial link between a genetic mutation and the dysfunctional behavior of microglial cells in the brain. This research sheds light on how expansions in the hexanucleotide repeat sequence of the C9orf72 gene—a known genetic culprit in ALS and frontotemporal dementia—disrupt the brain’s immune environment and impair microglial responses, potentially accelerating disease progression.
The intricate dance between neurons and microglia, the brain’s resident immune cells, is essential for maintaining neural health and responding to injury. However, in ALS patients harboring C9orf72 repeat expansions, this delicate balance is thrown into disarray. The study reveals that these expansions provoke a maladaptive microglial state characterized by impaired phagocytosis, altered inflammatory signaling, and an overall failure to mount an appropriate neuroprotective response.
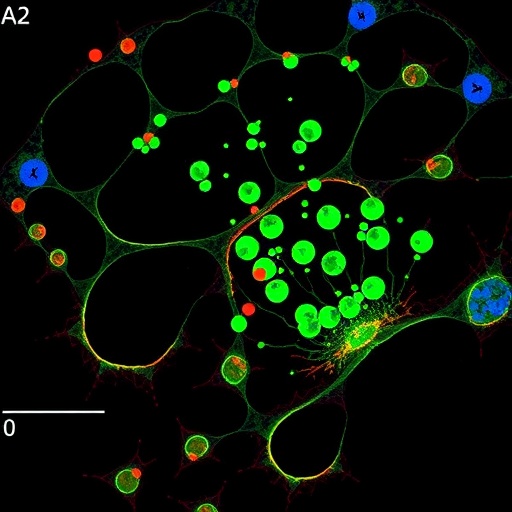
What sets this investigation apart is its multi-layered, methodical approach. By combining molecular profiling, advanced imaging techniques, and functional assays, the authors piece together a comprehensive picture of microglial dysfunction at a genetic, cellular, and systemic level. Their detailed analyses demarcate the sequence of microglial alterations triggered by C9orf72 repeats, providing insights into early pathogenic mechanisms that precede overt neurodegeneration.
One of the pivotal discoveries is the altered gene expression signature within microglia isolated from ALS patients with C9orf72 expansions. This signature reflects a shift away from homeostatic functions toward a phenotype marked by reduced ability to clear cellular debris and diminished capability to contain neuroinflammation. The ramifications of this shift are profound, as unchecked inflammation and impaired clearance mechanisms create a toxic milieu that exacerbates motor neuron vulnerability.
Key experimental models in this study employed patient-derived induced pluripotent stem cells (iPSCs) differentiated into microglia-like cells. This innovative platform enabled the team to observe how C9orf72-associated genetic anomalies manifest in microglial dysfunction in a controlled environment, free from confounding systemic variables. Additionally, the use of CRISPR-Cas9 genome editing to specifically manipulate C9orf72 repeats in these cells permitted direct causative links to be established.
Beyond molecular and cellular changes, the research also delves into the functional consequences of impaired microglial behavior. The investigators document a decreased capacity for phagocytosis—the process whereby microglia clear dead cells and harmful protein aggregates—alongside dysregulated secretion of cytokines and chemokines. These factors collectively fuel a pro-inflammatory state that not only fails to protect neurons but contributes actively to their demise.
The implications of such findings are wide-ranging. By unraveling how C9orf72 repeat expansions disrupt microglial function, this research opens new avenues for therapeutic interventions aimed at bolstering the brain’s innate immune defense. Modulating microglial activity could represent a strategy to slow or halt neurodegeneration in ALS, particularly in patients whose disease stems from this genetic mutation.
Moreover, the study engages with the broader context of neuroinflammation in neurodegenerative diseases. It highlights that microglial impairment may be a central driver in the pathology of ALS, reminiscent of mechanisms observed in Alzheimer’s and Parkinson’s diseases, but with distinct molecular signatures influenced by genetic background. This parallel suggests that targeting immune pathways could have cross-disease applicability.
The research team also explores the spatial and temporal dynamics of microglial changes within the brain. Using high-resolution imaging and tissue analysis from ALS patient samples, they reveal that microglial dysfunction is not uniform but varies across different neural regions. This spatial heterogeneity may explain why certain areas experience more pronounced degeneration, offering clues to disease progression patterns.
Importantly, the study addresses the chicken-and-egg question of whether microglial dysfunction precedes or follows neuronal damage. Their longitudinal data support the notion that impaired microglia contribute to early stages of ALS pathology, rather than merely responding to neuronal loss. This insight places microglia at the forefront of disease initiation and progression.
Another central theme of the paper is the role of C9orf72 protein itself in microglial biology. The authors provide evidence that loss-of-function effects caused by repeat expansions disrupt normal C9orf72 protein activities related to vesicle trafficking and autophagy—processes essential for microglial health and function. This mechanistic detail enriches our understanding of how genetic mutations translate into cellular dysfunction.
Beyond cellular and molecular mechanisms, the study’s findings resonate with clinical observations. Patients with C9orf72 expansions often exhibit a more aggressive disease course and distinct clinical features. Understanding how microglia contribute to this phenotype helps bridge the gap between genotype and disease manifestation, enhancing prospects for personalized medicine.
Future research, as outlined by the authors, will focus on identifying compounds and interventions capable of restoring microglial function in the context of C9orf72 expansions. Preclinical testing of immunomodulatory agents and gene-editing technologies may harness this knowledge to develop targeted therapeutics that could change the trajectory of ALS.
This study is poised to catalyze a paradigm shift in neurodegenerative disease research by emphasizing the centrality of microglial dysfunction intertwined with genetic mutations. The fusion of cutting-edge gene editing, cellular modeling, and patient-derived data forms a powerful nexus for innovation in understanding and eventually combating ALS.
In conclusion, this monumental work not only elucidates critical mechanisms by which C9orf72 hexanucleotide repeat expansions imperil microglial response but also provides a detailed roadmap for therapeutic targeting. These findings invigorate the field with hope that manipulating the brain’s immune landscape could offer a reprieve for patients suffering from one of neurology’s most devastating diseases.
Subject of Research: The study investigates how hexanucleotide repeat expansions in the C9orf72 gene impair microglial immune responses in ALS, elucidating the cellular and molecular mechanisms underlying neuroinflammation and neurodegeneration in this genetic form of the disease.
Article Title: C9orf72 hexanucleotide repeat expansions impair microglial response in ALS.
Article References:
Masrori, P., Bijnens, B., Fumagalli, L. et al. C9orf72 hexanucleotide repeat expansions impair microglial response in ALS. Nat Neurosci (2025). https://doi.org/10.1038/s41593-025-02075-1
Image Credits: AI Generated
Tags: advanced imaging techniques in neuroscienceC9orf72 gene mutationsfrontotemporal dementia connectionhexanucleotide repeat expansionsimmune response in ALSmicroglial cell behavior in brain healthmicroglial dysfunction in ALSmolecular profiling in ALS researchneuroinflammation in amyotrophic lateral sclerosisneuroprotective mechanisms in neurodegenerationpathogenic mechanisms in ALSphagocytosis impairment in microglia