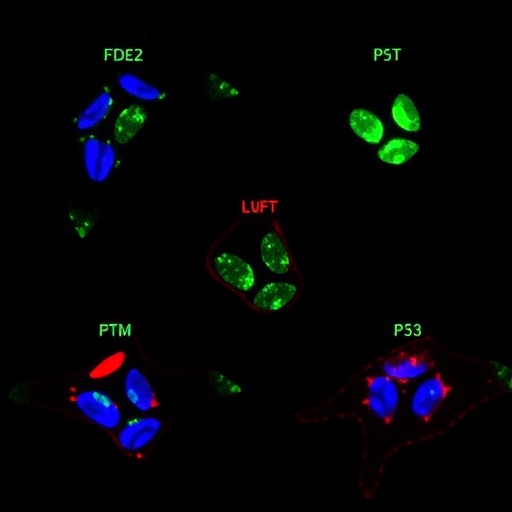
Cellular therapies have emerged as a transformative approach in the field of transplantation medicine, harnessing the bodyâs immune system to improve graft survival and combat disease. Among these therapies, regulatory T cell (Treg) therapies and virus-specific T cell (VST) therapies stand out as groundbreaking strategies that enhance the efficacy of transplantation while mitigating the risks of rejection and infection. This article explores the multifaceted roles of these therapies in transplantation, delineating their mechanisms of action, clinical applications, and future directions within the rapidly evolving landscape of cellular medicine.
Regulatory T cells, a specialized subset of T cells, play a crucial role in maintaining immune tolerance, preventing autoimmunity, and suppressing excessive immune responses. These cells are essential for the successful acceptance of transplanted organs and tissues. Tregs exert their immunosuppressive effects by inhibiting the activity of other immune cells, particularly effector T cells and B cells, thereby reducing inflammatory responses that could lead to graft rejection. In recent years, researchers have been investigating strategies to expand and manipulate Tregs ex vivo to enhance their therapeutic potential in transplantation settings.
One of the most promising avenues for Treg therapy involves the ex vivo expansion of these cells from the patient’s own blood. This approach minimizes the risk of graft-versus-host disease (GVHD), a common complication in stem cell and organ transplants in which the donor immune cells attack the recipient’s tissues. By expanding Tregs before transplantation, clinicians hope to create a robust population of suppressive cells that can effectively protect the graft without significantly hindering the patientâs immune response to pathogens.
Virus-specific T cell therapies have also gained attention as a potent strategy to combat viral infections that often complicate the post-transplant course. Patients undergoing transplantation are particularly susceptible to viral reactivations due to immunosuppression. The use of VSTs, which are customized T cells directed against specific viruses such as cytomegalovirus (CMV) or Epstein-Barr virus (EBV), has shown promising results in preventing or treating these infections. By rapidly expanding T cells that specifically target viral antigens, clinicians can provide a tailored immune response that helps control viral loads and reduce the incidence of associated complications.
Recent clinical trials have underscored the effectiveness of combining Treg and VST therapies in a unified treatment strategy. This dual approach allows for the maintenance of immune tolerance towards the transplanted organ while simultaneously bolstering the immune defense against common viral pathogens. The synergy between these two cellular therapies exemplifies the potential of personalized medicine in transplantation, where therapies are tailored to the specific needs of individual patients.
Moreover, research has highlighted the importance of the tissue microenvironment in shaping the functionality of Tregs and VSTs. The ability of these cells to migrate to the site of the graft, interact with other immune cells, and adapt to the local cytokine milieu is paramount to their success. Ongoing studies are elucidating the molecular signals and pathways that govern these processes, with the aim of refining therapeutic strategies to optimize Treg and VST function in the context of transplantation.
Notably, the development of gene-editing technologies such as CRISPR-Cas9 has opened new possibilities for enhancing cellular therapies. Researchers are investigating ways to genetically engineer Tregs and VSTs to augment their survival, functionality, and specificity. For instance, modifying Tregs to express certain cytokines or inhibitory receptors could enhance their ability to suppress unwanted immune responses in the transplant setting. Similarly, engineering VSTs to improve their proliferation and persistence could lead to more sustained antiviral responses, offering a compelling avenue for future research.
Despite these advancements, several challenges remain. The complexity of the immune system, coupled with inter-individual variability in immune responses, poses significant hurdles in developing universally effective therapies. Furthermore, the regulatory landscape for cellular therapies is evolving, as regulatory bodies seek to ensure safety and efficacy while fostering innovation. Navigating these complexities will require collaborative efforts between researchers, clinicians, and regulatory agencies to bring promising therapies from bench to bedside.
One of the most pressing concerns in cell-based therapies is the potential for unintended side effects. While Treg therapies aim to suppress harmful immune responses, there is a risk that they could also dampen the body’s ability to fight infections or tumors. Similarly, while VSTs are designed to target specific viral pathogens, the possibility of off-target effects must be carefully considered. To address these concerns, ongoing research focuses on the development of protocols for monitoring and managing adverse events associated with cellular therapies.
Public awareness and acceptance of cellular therapies also play a crucial role in their successful implementation. Education about the benefits and risks associated with these innovative approaches is essential for patients, families, and healthcare providers alike. Engaging diverse stakeholders in discussions about the promise of Treg and VST therapies can foster a more informed understanding and appreciation for these evolving treatment modalities.
As the field of cellular therapies in transplantation continues to advance, the integration of basic research and clinical practice will be critical. Robust clinical trials are necessary to evaluate the safety and efficacy of Treg and VST therapies across diverse patient populations and transplant types. As researchers uncover new insights into the immune system’s intricacies, these findings must be translated into practical applications that improve patient outcomes and revolutionize transplantation medicine.
In summary, regulatory T cell therapies and virus-specific T cell therapies represent the forefront of innovation in transplantation, offering new hope for improving graft survival and managing post-transplant complications. By harnessing the power of the immune system through personalized cellular therapies, researchers and clinicians are paving the way for more effective and safer transplantation options. The ongoing exploration of these therapies promises to transform the landscape of transplantation, ultimately leading to better outcomes for patients and greater longevity for transplanted organs.
In conclusion, the dynamic interplay between regulatory T cells and virus-specific T cells highlights the complexity of the immune response in the context of transplantation. Understanding and leveraging this interplay through cutting-edge research and clinical innovation will be paramount in overcoming the challenges that have long plagued transplantation medicine. The future of transplantation lies not only in organ procurement and surgical technique but also in the strategic use of cellular therapies to optimize outcomes for patients facing life-threatening conditions.
Subject of Research: Cellular Therapies in Transplantation
Article Title: Cellular Therapies in Transplantation â Regulatory T Cell Therapies and Virus Specific Therapies
Article References:
Hannouneh, Z.A., Merzkani, M., Hsieh, CS. et al. Cellular Therapies in Transplantation â Regulatory T Cell Therapies and Virus Specific Therapies.
Curr Transpl Rep 12, 33 (2025). https://doi.org/10.1007/s40472-025-00489-1
Image Credits: AI Generated
DOI:
Keywords: Cellular therapies, transplantation, regulatory T cells, virus-specific T cells, immune tolerance, graft survival.
Tags: cellular medicine innovationsclinical applications of T cell therapiescombating transplant rejectionenhancing graft acceptance through cellular therapiesex vivo expansion of Tregsfuture directions in transplantation medicinegraft survival improvementimmune tolerance in transplantationimmunosuppressive therapies in transplantationregulatory T cell therapiesT cell therapies in transplantationvirus-specific T cell therapies