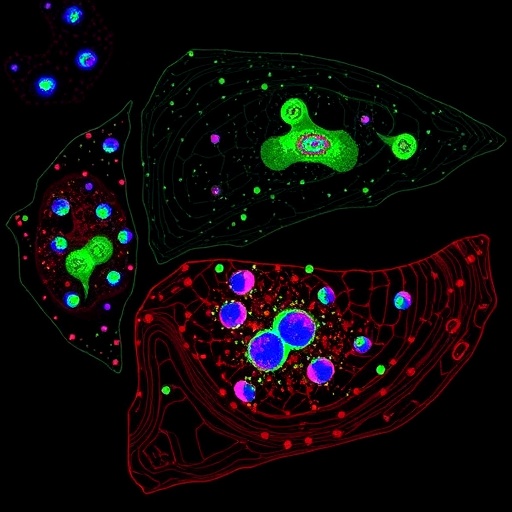
In a groundbreaking study published in Cell Death Discovery, a team of researchers led by Wang, K., Xiao, Y., and Wan, J. unveils an intricate metabolic-epigenetic axis that governs glioblastoma progression through the enzyme HADHA and its regulatory effects on the JAK/STAT3 signaling pathway. This discovery sheds new light on the cellular machinery driving one of the most aggressive brain tumors, offering promising avenues for targeted therapies that could revolutionize current treatment paradigms.
Glioblastoma multiforme (GBM) represents a dire medical challenge, notorious for its rapid proliferation, resistance to treatment, and dismal patient prognosis. Despite intensive research, effective targeted therapies remain elusive. The recent findings pinpoint the mitochondrial trifunctional protein subunit alpha (HADHA) as a pivotal metabolic regulator intricately linked to oncogenic signaling pathways involved in tumor survival and expansion. Such a dualistic function in both metabolism and epigenetic control is particularly compelling, bridging two formerly considered disparate realms of cancer biology.
HADHA’s canonical role involves the beta-oxidation of long-chain fatty acids within mitochondria, a critical component of cellular energy homeostasis. However, the novel insight from Wang and colleagues establishes a hitherto unknown function of HADHA in modulating JAK/STAT3 signaling—a pathway notoriously implicated in the proliferation, immune evasion, and stemness of glioblastoma cells. This dual functional capacity implies that metabolic enzymes may exert far-reaching influences beyond canonical bioenergetics, functioning as epigenetic modulators that sculpt oncogenic transcriptional programs.
.adsslot_vYLo7f2nM9{width:728px !important;height:90px !important;}
@media(max-width:1199px){ .adsslot_vYLo7f2nM9{width:468px !important;height:60px !important;}
}
@media(max-width:767px){ .adsslot_vYLo7f2nM9{width:320px !important;height:50px !important;}
}
ADVERTISEMENT
The study employs a sophisticated integration of metabolomic profiling, chromatin immunoprecipitation sequencing (ChIP-seq), and proteomic analyses to delineate how HADHA influences STAT3 phosphorylation and nuclear translocation. The data reveal that suppression of HADHA disrupts fatty acid oxidation flux, leading to alterations in the cellular acetyl-CoA pool. These metabolic changes then cascade to affect histone acetylation patterns, thereby epigenetically reprogramming STAT3 target gene expression. This mechanistic pathway suggests a feedback loop wherein mitochondrial metabolism directly informs chromatin architecture, fine-tuning gene expression landscapes critical for glioblastoma malignancy.
Importantly, the authors demonstrate that silencing HADHA expression in glioblastoma cell lines markedly diminishes tumor cell viability and invasiveness in vitro, effects that are rescued by enforced activation of STAT3 signaling. Such functional assays affirm the indispensable role of HADHA-mediated metabolic regulation in sustaining JAK/STAT3-driven oncogenic phenotypes. This crosstalk underscores an integrative axis that could be exploited pharmacologically; inhibiting HADHA might concurrently disrupt energy metabolism and epigenetic oncogene expression, delivering a one-two punch to tumor progression.
Further reinforcing clinical relevance, analysis of patient-derived glioblastoma specimens reveals a positive correlation between HADHA expression and STAT3 activation status, as well as poorer overall survival rates. These findings point toward HADHA not only as a mechanistic node but also as a prognostic biomarker for aggressive disease. The ability to stratify patients based on HADHA-STAT3 axis activity could refine precision oncology approaches and inform therapeutic decision-making.
Beyond glioblastoma, this study propels a paradigm shift concerning metabolic enzymes as epigenetic regulators. It embodies the concept that metabolism and gene regulation exist not as isolated processes but as deeply entwined networks that cooperate to drive tumor biology. By illuminating this previously unappreciated metabolic-epigenetic axis, the research opens fertile ground for investigating analogous pathways in other malignancies characterized by metabolic dysregulation and aberrant JAK/STAT signaling.
Equally significant is the methodological rigor with which the team interrogated the regulatory axis. Using CRISPR/Cas9-based genetic editing, targeted metabolite supplementation, and advanced microscopy techniques to visualize STAT3 localization changes, the research offers a multi-dimensional perspective. Such comprehensive approaches ensure that findings are not artifacts of in vitro models but robust phenomena with in vivo translational potential.
The therapeutic implications of these discoveries are vast. Traditional strategies targeting JAK/STAT pathways often encounter obstacles such as compensatory signaling and systemic toxicities. By targeting HADHA, an upstream metabolic regulator, there is potential to circumvent such resistance mechanisms while simultaneously impairing tumor energetics and epigenetic maintenance. Drug development efforts could focus on small molecules or peptides that specifically inhibit HADHA’s enzymatic function or disrupt its interaction with STAT3 co-factors, thus providing finely tuned interventions.
Moreover, combining HADHA inhibition with existing modalities such as temozolomide chemotherapy or immune checkpoint blockade may potentiate anti-tumor efficacy. Given the immunosuppressive microenvironment in glioblastoma, the ability to modulate metabolic-epigenetic pathways influencing immune evasion could reinvigorate host anti-tumor responses. Translational research aimed at evaluating such combinatorial regimens may pave the path for clinical trials.
Wang et al.’s elucidation of the HADHA-JAK/STAT3 axis underscores the growing appreciation for metabolic enzymes as versatile regulators that extend beyond their canonical functions. This blurring of boundaries between metabolism and epigenetics is emblematic of a new frontier in cancer biology, one that promises innovative therapeutic targets grounded in a nuanced understanding of tumor cell physiology.
In sum, the study’s revelations mark a transformative advance in glioblastoma research, offering a mechanistic blueprint for future interventions. The metabolic-epigenetic interplay mediated by HADHA and its impact on JAK/STAT3 signaling could redefine strategies aimed at combating this intractable cancer. As researchers continue to unravel the complexities of tumor biology, insights like these illuminate paths toward more effective, durable, and personalized treatments for patients facing the formidable challenge of glioblastoma.
Subject of Research: HADHA-mediated regulation of JAK/STAT3 signaling in glioblastoma through a metabolic-epigenetic axis
Article Title: HADHA-mediated regulation of JAK/STAT3 signaling in glioblastoma: a metabolic-epigenetic axis
Article References:
Wang, K., Xiao, Y., Wan, J. et al. HADHA-mediated regulation of JAK/STAT3 signaling in glioblastoma: a metabolic-epigenetic axis. Cell Death Discov. 11, 361 (2025). https://doi.org/10.1038/s41420-025-02660-0
Image Credits: AI Generated
DOI: https://doi.org/10.1038/s41420-025-02660-0
Tags: cancer metabolism and epigeneticsfatty acid beta-oxidation in cancerglioblastoma multiforme treatment challengesglioblastoma patient prognosisHADHA role in glioblastomainnovative glioblastoma therapiesJAK/STAT3 signaling pathwaymetabolic-epigenetic axis in cancermitochondrial trifunctional protein in tumorsoncogenic signaling in glioblastomatargeted therapies for brain tumorstumor survival mechanisms