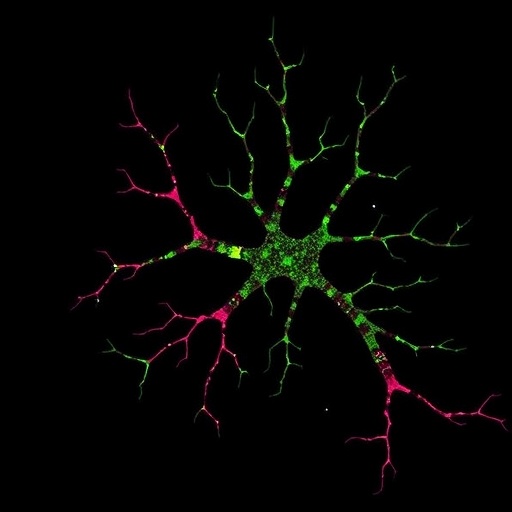
In the intricate architecture of the mammalian brain, the ability of cells to distinguish ‘self’ from ‘non-self’ is paramount for proper development and function. This intricate cellular self-recognition has been extensively documented in neurons, where it orchestrates neuronal self-avoidance, ensuring dendrites from the same neuron do not entangle but instead spread to optimize connectivity. The molecular underpinnings of this process have been illuminated by studies implicating clustered protocadherins (cPcdhs), a vast family of cell-surface adhesion molecules whose isoform diversity facilitates highly specific homophilic interactions. These interactions generate unique neuronal identities essential for the establishment of neural circuits during brain development. However, a critical question has persisted unresolved: could this self-recognition mechanism extend beyond neurons, perhaps influencing other glial cell types such as astrocytes?
A groundbreaking study by Lee et al., recently published in Nature, sheds light on this question by providing compelling evidence that astrocytes, the star-shaped glial cells integral to brain function, utilize a similar self-recognition mechanism to regulate their own morphogenesis. Central to this discovery is the gamma C3 (γC3) isoform of the Pcdhγ family, which the authors found to be selectively enriched in astrocytes from both human and mouse brains. This finding not only challenges the neuron-centric view of clustered protocadherin function but also broadens the biological significance of self-recognition molecules across diverse brain cell types.
Astrocytes are critical for maintaining neuronal health and synaptic activity, partly through their elaborate and highly branched processes that intimately associate with neurons and blood vessels. The developmental cues and molecular mechanisms sculpting astrocyte morphology have remained elusive, yet these physical architectures are essential for their ability to modulate neural circuits. By deploying a sophisticated genetic toolkit, Lee and colleagues dissected the role of γC3 in astrocytes within the mouse visual cortex, revealing that loss of γC3 impairs the normal morphological development of these cells. Their approach involved creating γC3-null astrocytes and assessing how the absence of this isoform affects cellular shape and process elaboration at the microscopic level.
.adsslot_bCzdOGE2rL{width:728px !important;height:90px !important;}
@media(max-width:1199px){ .adsslot_bCzdOGE2rL{width:468px !important;height:60px !important;}
}
@media(max-width:767px){ .adsslot_bCzdOGE2rL{width:320px !important;height:50px !important;}
}
ADVERTISEMENT
The investigation advanced further by addressing whether γC3 operates through a homophilic interaction mechanism analogous to that observed in neurons. In neuronal self-avoidance, thousands of cPcdh isoforms mediate highly selective homophilic binding — a lock-and-key mechanism by which processes from the same neuron recognize and repel each other to avoid overlapping. To parse out if the same kind of interaction occurs in astrocytes, the authors ingeniously engineered chimeric γC3 proteins designed to heterophilically bind to one another but incapable of homophilic binding. This molecular innovation allowed them to uncouple the typical homophilic recognition from novel heterophilic interactions, providing a powerful tool to test the necessity and sufficiency of specific binding patterns in astrocyte morphology.
Remarkably, astrocytes that co-expressed complementary heterophilic γC3 chimeras restored their normal morphological features despite lacking the capacity for traditional homophilic interactions. Conversely, expression of either chimeric protein alone in γC3-null astrocytes failed to rescue morphology, highlighting that astrocyte morphogenesis depends on self-recognition via matching γC3 molecules on the same cell. These results establish that astrocytic self-recognition does not merely involve passive adhesion but an active recognition mechanism critical for shaping the astrocyte’s elaborate architecture.
This discovery adds a novel dimension to our understanding of astrocyte biology. While astrocytes have long been recognized for their supportive and regulatory roles within the central nervous system, their self-recognition capabilities open new avenues for exploring how intrinsic molecular codes govern glial cell organization and networking. Moreover, the parallels drawn between neuronal and astrocytic self-avoidance mechanisms suggest a conserved evolutionary strategy wherein diversified cadherin isoforms encode cellular identity and spatial patterning across distinct cell types in the brain.
The implications extend beyond fundamental neurobiology. Precise astrocyte morphogenesis is known to influence synaptic formation and plasticity, neurovascular coupling, and metabolic support to neurons. Any perturbation in astrocyte shape or function can have cascading effects on overall brain circuitry and potentially contribute to neurological disorders. Understanding the molecular signals that guide astrocyte self-recognition, such as γC3-mediated homophilic binding, provides potential targets for therapeutic intervention in diseases characterized by glial dysfunction or aberrant neural connectivity.
Methodologically, the study stands out for its innovative design in generating γC3 chimeric proteins with altered binding specificity. This approach exemplifies how molecular engineering can dissect complex cell-cell recognition systems that depend on subtle variations in isoform expression. Such precision tools enable not only mechanistic insights but also could serve as templates for future manipulation of cell recognition in regenerative medicine or synthetic biology applications.
Furthermore, the observed enrichment of γC3 in both human and mouse astrocytes underscores the translational relevance of these findings. It suggests that the molecular frameworks controlling astrocyte morphogenesis are highly conserved, warranting deeper exploration of cPcdh diversity in human brain development and pathology. This insight also encourages revisiting previous models of astrocyte development under the lens of isoform-specific self-recognition, which might reconcile divergent observations in the field.
The study by Lee et al. also revitalizes the discussion of glial cell autonomy. While historically overshadowed by neurons, astrocytes and other glia are increasingly recognized as active participants in neural circuit formation. The evidence that astrocytes utilize protocadherin-mediated self-recognition to direct their own morphology affirms their role as intrinsic architects of the neural microenvironment, sculpting the brain’s cellular landscape through autonomous molecular codes.
In sum, this research articulates a remarkable expansion of the self-recognition paradigm from neurons to astrocytes, anchored by the pivotal role of the γC3 protocadherin isoform. It opens new horizons for understanding brain development and points to the pervasive influence of molecular identity codes across cell types. As the field advances, probing the combinatorial expression and interaction of clustered protocadherins in diverse glial populations may reveal an intricate molecular tapestry that governs brain organization at multiple levels.
The findings reported set a foundation for novel investigations into astrocyte-neuron interactions, highlighting how self-recognition mechanisms might govern not only intra-cellular patterning but also intercellular communication within the neural milieu. Ultimately, unraveling these processes holds promise for deciphering the cellular basis of cognition and neurological disease, offering paths toward innovative therapeutic strategies rooted in cellular identity and recognition.
Subject of Research: Astrocyte morphogenesis and self-recognition mediated by γC3 protocadherin isoform
Article Title: Astrocyte morphogenesis requires self-recognition
Article References:
Lee, J.H., Sergeeva, A.P., Ahlsén, G. et al. Astrocyte morphogenesis requires self-recognition. Nature (2025). https://doi.org/10.1038/s41586-025-09013-y
Image Credits: AI Generated
Tags: astrocyte identity and functionastrocyte morphogenesisastrocytes in neural circuitsbrain architecture and connectivitycell self-recognition mechanismsgamma C3 isoform significanceglial cell developmentimplications of astrocyte self-recognitionmolecular interactions in glianeurodevelopmental processesneuronal self-avoidanceprotocadherins in brain function