Scientists at the University of Stuttgart have made groundbreaking advancements in the realm of synthetic biology through their innovative application of DNA nanotechnology. Under the leadership of Professor Laura Na Liu, this research team has been able to deftly control the structure and functionality of biological membranes using cleverly designed “DNA origami”. This new system has the potential to transform how therapeutic loads are delivered into cells, marking a major leap forward in targeted medication administration and other crucial therapeutic interventions. The findings have been documented in the prestigious journal Nature Materials, showcasing a pioneering approach that could redefine therapeutic delivery mechanisms.
The research hinges on an important realization: the shape and morphology of a cell fundamentally influence its biological functions. This notion, intimately known as “form follows function,” echoes throughout various disciplines, including modern design and architecture. However, transferring this principle to the realm of synthetic cells has posed significant challenges. Yet, advancements in DNA nanotechnology are now presenting promising solutions, enabling the innovative construction of transport channels that facilitate the movement of large therapeutic proteins across cell membranes. By harnessing the capabilities of DNA origami, Liu’s team has pioneered a technique that holds enormous promise for advancing the field of synthetic biology.
A major milestone has been achieved with the development of a groundbreaking tool that allows for the precise regulation of the shape and permeability of lipid membranes within synthetic cells. These membranes are created using lipid bilayers that enclose an aqueous region, serving the purpose of simplified models of biological cells. These membranes have become invaluable for investigating the dynamics of membranes, understanding protein interactions, and exploring the behavior of lipids. The work of Liu and her team stands to significantly influence the research landscape in therapy development by providing a reliable method for manipulating the behavior of synthetic cells.
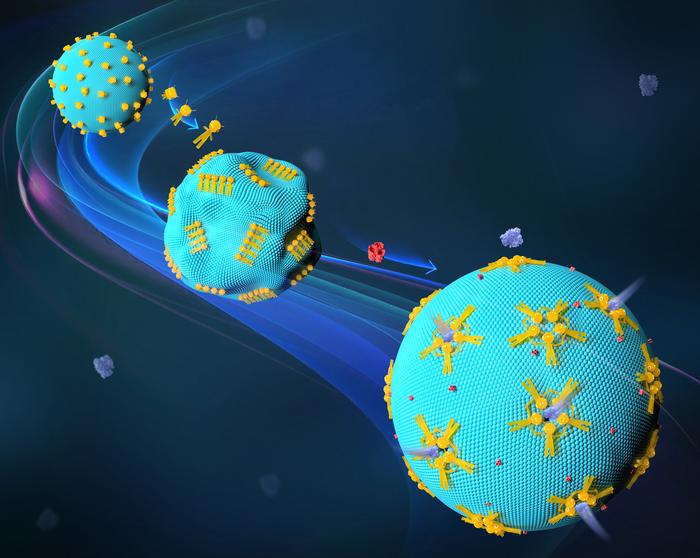
One key aspect of their research involves giant unilamellar vesicles (GUVs), which are engineered to mimic living cells due to their size and structure. The team employed signal-responsive DNA nanorobots, programming them to interact with these synthetic cells in controllable and predictable manners. This innovative approach has allowed researchers to effectively alter the shape and functionality of these GUVs—an achievement Liu refers to as a revolutionary milestone in the practical application of DNA nanotechnology.
Moreover, the new DNA nanorobots have shown the ability to create transport channels for proteins and enzymes as they navigate through synthetic cellular structures. These DNA origami constructs are designed to respond dynamically to their environment, enabling them to change shape and exert influence over their immediate surroundings on a micro meter scale. When the researchers coupled the transformations of the DNA nanorobots with the deformation of GUVs, they successfully engineered synthetic channels capable of letting large molecules traverse the membranes, while also possessing the ability to reseal as required.
The implications of this work are extensive and thought-provoking. One particularly exciting notion is that these DNA nanorobots can shape and configure GUVs to form transport channels within the membranes. Such channels could potentially facilitate the passage of therapeutic proteins when applied in living cells, providing the ability to transport critical medical interventions directly to their targets. In the landscape of synthetic biology, this poses a fascinating question: Could these synthetic platforms exemplify simplicity while maintaining functionality in biological settings, ultimately advancing both research and therapeutic practices?
Not only does this research open the door to enhanced delivery mechanisms for therapeutic agents, but it also proposes a leap forward in understanding the mechanisms of disease and crafting improved therapies. The DNA nanorobots’ capacity for creating cross-membrane channels enhances molecular transport efficiency, a critical factor when considering drug delivery systems. The channels can be engineered to be large enough to allow the passage of bulky therapeutic proteins or enzymes, transforming how treatments can be administered within living systems. The potential for closing these channels on command adds another layer of versatility, paving the way for more sophisticated therapeutic strategies.
As the synergy between DNA nanotechnology and synthetic cellular platforms grows, significant inquiries emerge. Researchers pose substantial questions regarding the simplicity of design versus effective complex biological functions. For instance, could artificial constructs simplify complexity while still being effective within a living biological environment? This study, among others, aims to further explore these innovative paradigms.
The contributions of Liu and her team, comprised of experts from the 2nd Physics Institute, the Department of Biophysics at the Institute of Biomaterials and Biomolecular Systems, and the Institute of Theoretical Physics IV at the University of Stuttgart, collectively strengthen the foundation of their groundbreaking research. This interdisciplinary approach enables the integration of various scientific perspectives and methodologies, further enriching the potential advancements in the field. Among the co-authors is Professor Hao Yan, a notable recipient of the Alexander von Humboldt Research Award, who contributes his substantial expertise from Arizona State University.
By effectively utilizing advanced techniques to manipulate synthetic cellular systems, the team’s findings promise to be at the forefront of a revolution in therapeutic delivery. The capabilities unveiled through their research might ultimately emulate biological systems, offering new methods to interact with living cells effectively. As therapeutic strategies evolve to utilize these sophisticated tools, the implications on health care and disease treatment could be profoundly transformative.
Furthermore, this pioneering study is not just a testament to what has been achieved but a clarion call for future research in this burgeoning field. Morphological transformation and the formation of channel systems stand to benefit diverse applications such as targeted drug delivery, biosensing, and even regenerative medicine. As the scientific community delves deeper into the intricacies of DNA-nanotechnology interfaces, the potential to model and manipulate biological mechanisms instills both excitement and anticipation for advancements that can significantly impact medical science and therapeutic practices.
As research continues to scale new heights, there is palpable anticipation surrounding the advancements that synthetic biology holds. The integration of DNA nanotechnology into therapeutic frameworks suggests a promising future for how complex biological systems can be understood and replicated for enhanced health outcomes. Each step forward offers not just a glimpse into improved treatments but opens the door to entirely new paradigms in how we approach medicine, disease, and healing.
This landmark study by Liu and her colleagues encapsulates the essence of pioneering scientific inquiry—melding creativity with discipline, rigor with innovation. The road ahead may still hold countless mysteries, yet with each discovery comes the exhilarating promise of redefining what is possible within the vast landscape of health interventions.
Subject of Research:
Article Title: Morphological transformation and formation of membrane channels in synthetic cells via reconfigurable DNA nanorafts
News Publication Date: 13-Jan-2025
Web References: Nature Materials
References: DOI: 10.1038/s41563-024-02075-9
Image Credits: University of Stuttgart / 2nd Physics Institute
Keywords: DNA nanotechnology, synthetic biology, therapeutic delivery, DNA origami, cell membranes, synthetic cells, membrane channels, drug administration, medical technology, molecular biology, innovative therapies, biological frameworks.