Toxic heavy metals found in wastewater have health and safety ramifications for communities affected by pollution. Hexavalent chromium is a dangerous, cancer-causing byproduct of industrial processes that is known to cause birth defects, severe diarrhea, and is linked to kidney, bladder, and liver cancers. Famously, it was the center of the lawsuit dramatized in the film “Erin Brockovich.”
Credit: Polyoxometalates, Tsinghua University Press
Toxic heavy metals found in wastewater have health and safety ramifications for communities affected by pollution. Hexavalent chromium is a dangerous, cancer-causing byproduct of industrial processes that is known to cause birth defects, severe diarrhea, and is linked to kidney, bladder, and liver cancers. Famously, it was the center of the lawsuit dramatized in the film “Erin Brockovich.”
Researchers are trying to find effective ways to remove hexavalent chromium from wastewater and a recently published paper shows how photocatalytic technology may be a solution. Photocatalysis is when light and a catalyst are used to speed up chemical reactions.
The paper was published in Polyoxometalates on March 23.
“Rapid industrialization causes an increased release of wastewater containing heavy metal ions. Hexavalent chromium, which has high carcinogenicity and teratogenicity, is widely found in wastewater and can easily enter food chains,” said Yuan-Yuan Ma, a researcher at Hebei Normal University in Shijiazhuang, China. Photocatalysis technology is an appealing solution for removing heavy metals from wastewater because it is sustainable, cost-effective, and environmentally friendly.
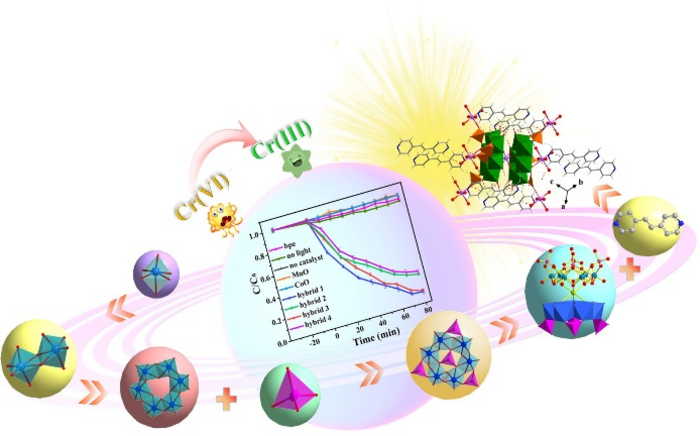
“This green approach for the removal of heavy metal ions uses sustainable light energy via hourglass-type phosphomolybdate-based crystalline photocatalysts and develops a strategy for the regulation of photocatalytic performance by adjusting the central metal ions in hourglass-type phosphomolybdate clusters,” said Ma. Researchers chose this particular type of photocatalyst because of its molecular properties and well-defined hourglass-type structure, which give it a wide light absorption ability and the band structure necessary to reduce the levels of hexavalent chromium.
Researchers tested four “hybrid” photocatalysts and compared them to six other photocatalysts. The hybrids had slightly different compositions, but all had the same hourglass-type structure that could be maintained up to 200 degrees Celsius. They had wide visible-light absorption and similar energy band structures. Researchers labeled these as Hybrid 1, 2, 3, and 4. After 80 minutes of exposure to a 10W LED light, hybrid 1 and 3 had around a 90% reduction in hexavalent chromium, while 2 and 4 had around a 74% and 71% reduction in hexavalent chromium respectively.
The hybrids generally performed better than any of the tested photocatalysts. Hybrids 1 and 3, which performed best, both were Mn{P4MO6}2-based hybrids. Hybrids 2 and 4 were Co{P4MO6}2-based. Researchers suspect that the better performance was due to structural differences that gave hybrids 1 and 3 a narrower band gap. “The semiconductor photocatalysts in photocatalytic processes can absorb photons matched with their band gap energy, leading to the conversion of toxic hexavalent chromium to less toxic chromium,” said Ma.
Looking ahead, researchers will focus on improving the design of the photocatalysts, while also planning for how to best bring this technology to a real-world wastewater setting. “Designing effective photocatalysts is the first step to solve heavy metal pollution in water,” said Ma. “We will design more efficient photocatalysts and apply the developed photocatalysts to actual industrial wastewater. We will also expand the treatment range of polluted water sources and strive to realize the practicality of the photocatalyst materials used.”
Other contributors include Xiao-Yu Yin, Hao-Xue Bi, Hao Song, Jing-Yan He, Ting-Ting Fang, and Zhan-Gang Han of Hebei Normal University.
The National Natural Science Foundation of China, the Natural Science Foundation of Hebei Province, the Science and Technology Project of Hebei Education Department, the China Postdoctoral Science Foundation Funded Project, the Science Foundation of Hebei Normal University, the Innovation Capability Improvement Plan Project of Hebei Province, and the College Student Science and Technology Innovation Project of Hebei Province supported this research.
##
About Polyoxometalates
Polyoxometalates is a peer-reviewed, international and interdisciplinary research journal that focuses on all aspects of polyoxometalates, featured in rapid review and fast publishing, sponsored by Tsinghua University and published by Tsinghua University Press. Submissions are solicited in all topical areas, ranging from basic aspects of the science of polyoxometalates to practical applications of such materials. Polyoxometalates offers readers an attractive mix of authoritative and comprehensive Reviews, original cutting-edge research in Communication and Full Paper formats, Comments, and Highlight.
About SciOpen
SciOpen is a professional open access resource for discovery of scientific and technical content published by the Tsinghua University Press and its publishing partners, providing the scholarly publishing community with innovative technology and market-leading capabilities. SciOpen provides end-to-end services across manuscript submission, peer review, content hosting, analytics, and identity management and expert advice to ensure each journal’s development by offering a range of options across all functions as Journal Layout, Production Services, Editorial Services, Marketing and Promotions, Online Functionality, etc. By digitalizing the publishing process, SciOpen widens the reach, deepens the impact, and accelerates the exchange of ideas.
Journal
Polyoxometalates
DOI
10.26599/POM.2023.9140027
Article Title
Photoactive hourglass-type M{P4Mo6}2 networks for efficient removal of hexavalent chromium
Article Publication Date
23-Mar-2023