In a groundbreaking study set to reshape our understanding of cardiac immune responses, researchers have uncovered the pivotal role of β2 adrenergic receptors in directing neutrophil behavior following myocardial infarction. This discovery sheds light on the intricate molecular mechanisms that orchestrate immune cell dynamics in ischemic cardiac tissue, holding promising implications for therapeutic strategies aimed at mitigating heart damage after an infarct.
Myocardial infarction, commonly known as a heart attack, triggers a cascade of inflammatory responses as the body’s immune system rushes to repair the damaged heart muscle. Central to this process is the mobilization of neutrophils—white blood cells that serve as the first responders to tissue injury. Until now, the precise molecular pathways guiding the recruitment and behavior of neutrophils in the ischemic heart remained elusive, impeding the development of targeted interventions.
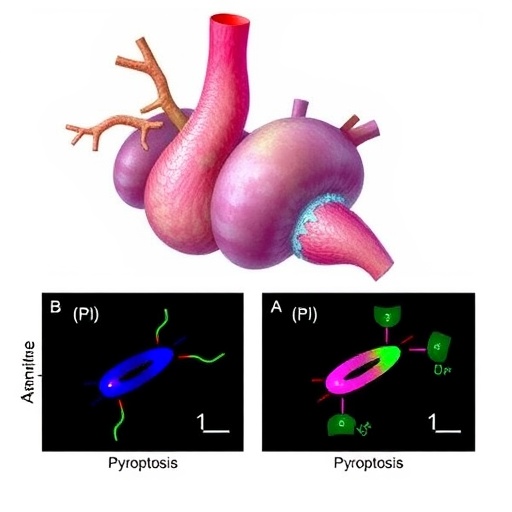
The study pinpointed the β2 adrenergic receptor (β2AR), a G protein-coupled receptor primarily recognized for its role in sympathetic nervous system signaling, as a key regulator influencing neutrophil demargination and homing to the infarcted myocardium. Demargination refers to the process by which neutrophils detach from the vascular endothelium—a critical step enabling them to enter circulation and subsequently infiltrate injured tissues.
Employing state-of-the-art in vivo imaging and molecular biology techniques, the research team demonstrated that activation of β2AR on neutrophils triggers intracellular signaling cascades leading to cytoskeletal reorganization and enhanced motility. This receptor-mediated modulation enables neutrophils to disengage from the vessel walls, facilitating their recruitment to regions of cardiac ischemia.
The findings challenge previous paradigms that predominantly attributed neutrophil trafficking to cytokine and chemokine gradients, introducing a novel dimension centered on adrenergic receptor signaling. The sympathetic nervous system, activated during acute stress and injury, appears to directly influence immune cell dynamics via β2AR engagement, thus integrating neural and immune responses in the context of myocardial infarction.
Importantly, the study elucidates that pharmacological manipulation of β2AR activity can significantly modulate the extent of neutrophil infiltration in the heart. Using selective β2AR agonists and antagonists in murine infarction models, the researchers observed marked differences in neutrophil accumulation and subsequent tissue damage. These data suggest therapeutic avenues whereby fine-tuning β2AR signaling might mitigate excessive inflammation and preserve myocardial function.
Delving deeper into molecular mechanisms, the research identified downstream effectors of β2AR activation, including cyclic AMP (cAMP) production and protein kinase A (PKA) signaling. These molecules orchestrate changes in neutrophil adhesion molecule expression and motility, effectively coordinating the demargination process. Such mechanistic insights provide a framework for developing drugs that target specific nodes in the signaling network to achieve precise immunomodulation.
The temporal dynamics of neutrophil recruitment also emerged as a critical factor influenced by β2AR signaling. Early engagement of these receptors post-infarction accelerates neutrophil mobilization, implicating a time-sensitive window during which therapeutic interventions could yield maximal benefit. This highlights the necessity for rapid clinical assessment and targeted treatment following cardiac ischemic events.
Beyond the immediate implications for myocardial infarction, the study’s revelations about β2AR’s role in immune cell trafficking may extend to other pathologies characterized by acute inflammation and tissue injury. Conditions such as stroke, sepsis, and autoimmune diseases may similarly involve adrenergic receptor-mediated modulation of leukocyte behavior, broadening the impact of this research across multiple disciplines.
The integration of neural and immune system responses via β2AR signaling underscores an emerging conceptual framework in immunology, emphasizing the bidirectional communication between these systems. Understanding how the nervous system influences immune cell function opens exciting possibilities for neuroimmune therapeutics aimed at restoring homeostasis in diverse disease contexts.
This research also poses intriguing questions about the potential side effects of β2AR-targeted therapies currently employed in treating cardiovascular and pulmonary disorders. The modulatory role of β2AR in neutrophil mobilization suggests that existing drugs could inadvertently influence immune responses, necessitating careful evaluation of their immunological repercussions.
Future investigations are poised to dissect the interplay between β2AR and other receptor systems governing neutrophil dynamics, such as chemokine receptors and integrins. Elucidating how these pathways converge to fine-tune neutrophil recruitment and activity will deepen our understanding of inflammatory regulation and aid in designing multifaceted therapeutic approaches.
Moreover, the identification of β2AR as a central player invites exploration into patient-specific factors influencing receptor expression and responsiveness. Genetic polymorphisms, comorbidities, and environmental factors may modulate β2AR-mediated neutrophil behavior, offering opportunities for personalized medicine tailored to individual immunological landscapes.
In conclusion, the discovery of β2 adrenergic receptors as orchestrators of neutrophil demargination and recruitment to the ischemic heart marks a significant advance in cardiovascular immunology. By unveiling the molecular dialogues between the nervous and immune systems that mobilize frontline defenders to sites of cardiac injury, this work paves the way for innovative treatments aimed at balancing protective inflammation with tissue preservation. As research progresses, harnessing the power of β2AR signaling could revolutionize how we manage myocardial infarction and other inflammatory disorders, heralding a new era of targeted immunomodulation.
Subject of Research: Role of β2 adrenergic receptors in neutrophil demargination and recruitment following myocardial infarction.
Article Title: β2 adrenergic receptors orchestrate neutrophil demargination and recruitment to the ischemic heart following myocardial infarction.
Article References: Dahdah, A., Maremanda, K.P., Marimuthu, M. et al. β2 adrenergic receptors orchestrate neutrophil demargination and recruitment to the ischemic heart following myocardial infarction. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71612-8
Image Credits: AI Generated
Tags: G protein-coupled receptors in heart attackimmune cell dynamics in heart repairin vivo imaging of neutrophil activityischemic cardiac tissue inflammationmolecular mechanisms of neutrophil recruitmentneutrophil behavior after myocardial infarctionneutrophil demargination processneutrophil homing to injured myocardiumsympathetic nervous system and immune modulationtargeted therapies for post-infarct inflammationtherapeutic strategies for myocardial infarctionβ2 adrenergic receptors in cardiac immune response